Feed Regulatory Renewal Consolidated Modernized Framework Proposal – November 2015
This page has been archived
Information identified as archived is provided for reference, research or record-keeping purposes. It is not subject to the Government of Canada Web Standards and has not been altered or updated since it was archived. Please contact us to request a format other than those available.
Table of Contents
- Purpose
- Objectives
- Overview of the Proposed Regulatory Changes
- Detailed Discussion of the Proposed Regulatory Framework
- Risk-based Oversight
- Support for Small Businesses
- Expected Coming into Force of the Proposed Regulations
- Guidance Documents and Model Systems
- User Fees Modernization
- Conclusion
- Next Steps
- Annex 1: What We Heard from Consultations
- Annex 2: Proposed Labelling Requirements
- Annex 3: Written PCP
Purpose
Following extensive stakeholder pre-consultation in 2012, the CFIA developed and released a series of subject-specific proposals for public review and comment regarding the modernization of Canada's federal feed regulatory framework:
- Feed ingredient Assessment & Authorization (Completed – Consultation closed December 2013);
- Feed Labelling (Completed – Consultation closed December 2013); and
- Feed Hazard Identification and Preventive Controls (Completed – Consultation closed July 25, 2014)
As a next step in its regulatory development process, the CFIA has prepared this Consolidated Proposal for further consultation with stakeholders such as suppliers of feed ingredients, commercial feed mills, feed distributors and feed retailers, livestock producers, industry associations, other government departments and international trading partners. The proposal seeks to:
- Integrate the first three modules to demonstrate how they will work together to provide a robust, risk-based regulatory framework;
- Add information on facility permissions (licensing/inspection/enforcement) to demonstrate how the principles of the modernized Integrated Agency Inspection Model (iAIM) will apply in a feed context; and
- Propose additional key regulatory requirements not addressed in the proposals/consultations to date.
On the basis of feedback received from consultation on these proposals, the CFIA will prepare a comprehensive regulatory proposal for publication in the Canada Gazette Part I in the coming months.
Objectives
At a corporate level, the CFIA has embarked on a comprehensive change agenda to strengthen its foundation of legislation, regulatory programs, and inspection delivery with the following goals:
- Legal frameworks that are robust, flexible, and consistent with international approaches and appropriate for 21st century;
- Regulations that reduce unnecessary compliance burden and support innovation while maintaining public safety, as well as environmental and economic sustainability; and
- Inspection delivery that is based on common inspection activities and standard processes, and supported by a renewed training regime.
With respect to the Agency's livestock feed regulatory program, the achievement of these goals is being realized:
- Amendments to modernize the Feeds Act were tabled in Parliament in December 2013 as part of the Agricultural Growth Act, a comprehensive bill that aimed to modernize several statutes administered by the CFIA. The Agricultural Growth Act and the statutory amendments it contained in it received Royal Assent and came into force in late February 2015.
- As part of a comprehensive, multi-year regulatory modernization process, the CFIA launched its Feed Regulatory Renewal project in January 2012 as one of several priority frameworks for modernization; and
- Following extensive development and consultation during 2012-2014, the CFIA has finalized a modernized, Integrated Agency Inspection Model (iAIM) that is being implemented across all Agency food, animal and plant health regulatory programs.
In initiating the Feed Regulatory Renewal project in 2012, the following project-specific objectives have formed the basis for proposals and consultation to date:
Develop a modernized risk-based and outcome-based regulatory framework for feeds which:
- safeguards feeds and the food production continuum;
- attains the most effective and efficient balance between fair and competitive trade in the market; and
- minimizes regulatory burden.
In bearing these objectives in mind, this proposal also identifies how both the Feeds Act amendments provided by the Agricultural Growth Act and the iAIM are influencing the design of a modernized regulatory framework for livestock feed.
Overview of the Proposed Regulatory Changes
Synopsis
The modernized regulatory framework proposes to:
- Build a stronger, more transparent and flexible feed ingredient assessment and authorization process as the basis for a safe and competitive feed supply chain;
- Enable the consideration and adoption of ingredient authorization processes and decisions of other jurisdictions to reduce burden and promote market access;
- Update the positive list of authorized feed ingredients and capitalize on new legal authorities to improve timeliness and accuracy so ingredient definitions reflect current characteristics, science and technology;
- Reduce the number of feeds that will require mandatory pre-market registration;
- Adopt an outcome-based and systems-based approach to feed safety and compliance which will require regulated parties to conduct hazard identification and put in place preventive controls to address risks posed by any identified hazards. An outcome-based approach allows for the application to a wide range of feeds and processes and to reflect differences in the size of businesses and processing complexity;
- Increase flexibility in labelling requirements;
- Implement a risk-based permissions (licencing/registration) regime for individuals or establishments and traceability requirements to better support risk management along the feed supply chain and timely response to incidents where risks to public, animal or plant health or the environment is required;
- Enhance the oversight of feed imports and exports to improve alignment with domestic requirements.
Other aspects of the regulations, such as standards, will be updated to better reflect current science, production practices and technology.
Canada's current feed regulations are focused on setting standards for feeds and controlling risks via product registration and other forms of pre-market assessment and approval. The Feed Regulatory Renewal project, taking into account new authorities provided by the Agricultural Growth Act and other Canadian Food Inspection Agency (CFIA) transformation initiatives, represents an opportunity for Canada to fundamentally change its regulatory framework for feed control and oversight and to continue to position Canada as a leader in feed safety and incident response.
The proposed regulations will reflect internationally recognized standards and management-based requirements, including good manufacturing practices and the Codex Code of Good Animal Feeding Practices. The feed chain as a whole will need to take on this preventive control approach to address hazards before they become a problem. The proposed regulations will also reflect the guiding principles of the CFIA's modernized, Integrated Agency Inspection Model (iAIM), developed following extensive consultations in 2012 and 2013, and support its implementation.
The proposed regulatory framework would further:
- enable regulated parties to rapidly mitigate emerging feed and food safety risks;
- confirm industry responsibility and accountability for preparing safe feed;
- enable the CFIA to apply a consistent science and risk-based approach to oversight;
- reduce unnecessary regulatory burden on stakeholders;
- reflect changes in science and technology;
- support innovation; and,
- address other regulatory requirements in a more current, consistent manner.
The proposed regulations will be supplemented by a suite of new guidance documents that outline the intent of the requirements and will assist regulated parties in complying with the requirements, along with model systems that have been demonstrated to achieve compliance when properly applied. The CFIA will release examples of guidance documents and plain language model systems developed to assist small businesses to meet the requirements and intends to consult with industry to determine how best to draft guidance documents to help industry achieve regulatory compliance.
Highlights
The following sections provide an overview of the key elements of the proposed framework.
With respect to the Modernized Regulatory Requirements:
- The scope of the species to which the Feeds Regulations would apply would be expanded to include feeds for animals that are raised for human consumption but are not currently regulated, including cervids (e.g., deer, elk), bison and honeybees;
- The range of existing product-oriented standards for feeds, including safety standards, will be reviewed, revised, reduced or expanded as appropriate to address current regulatory objectives (outcome-, risk-based and systems-based approaches) and expected outcomes;
- new authorities provided by the Agricultural Growth Act will also be taken advantage of to incorporate technical and other standards by reference wherever possible (e.g. positive list of authorized ingredients) to support more frequent and timely updating;
- Hazard identification and assessment would be required by all those involved throughout the feed supply chain with respect to the feed-related activities in which they are involved;
- Following the identification and assessment of hazards, a preventive control plan (PCP) would be required from any regulated party that manufactures or sells feed domestically or imports or exports feeds;
- Labelling requirements, would provide additional flexibility while adding elements for safety and traceability; and
- International standard for traceability established by Codex Alimentarius would be applied to all feed manufacturers, licence holders and persons importing, exporting or trading feeds domestically.
With respect to Permissions (authorizations, registrations and licences):
- A robust, more risk-based and transparent ingredient assessment and authorization process would be implemented:
- opportunities to accept application packages and/or foreign country authorizations for new ingredients will be examined where the foreign country's feed ingredient approval process meets Canadian requirements;
- A reduction in the kinds of mixed feeds that would require pre-market registration is proposed; for reasons of risk, only milk replacers, feeds administered via water, medicated mineral feeds and flavouring agents of any origin (domestically-produced or imported) would be subject to mandatory registration; and
- New authorities provided by the Agricultural Growth Act would be activated to require persons or establishments to obtain permissions (licences or registrations) to engage in inter-provincial, import and export trade activities.
Sector-by-Sector Anticipated Impacts
Domestic Trade
Ingredient Manufacturers/Suppliers
- New feed ingredients must be assessed and authorized by CFIA before they can be manufactured or sold for use in livestock feed.
- Submissions for new ingredient authorization will require data to support the safety of the ingredient as well as information to support the feed purpose for the ingredient.
- The CFIA will collaborate with Health Canada and industry to develop and maintain a list of permissible claims that can be used on certain ingredient labels without need for pre-market approval.
- Authorized ingredients for sale or distribution in Canada must comply with standards and labelling requirements for the ingredient as set out in the Regulations (including new requirements for traceability) and CFIA's positive list of authorized feed ingredients (currently Schedules IV and V of the Regulations)
- Clarity on requirements for the use of global (multijurisdictional) labels on feed ingredients to be included in modernized regulations.
- Ingredient manufacturers will be required to perform a hazard analysis and create and implement a written preventive control plan to address risks to public, animal or plant health or the environment posed by the hazards.
- Sector-specific programs that include compliance with feed regulatory requirements as a specification may be considered as Model Systems and be taken into consideration by the CFIA in the design and delivery of risk-based inspection oversight programs.
- A permission may be required where ingredients are sold or distributed inter-provincially.
- Some ingredients may require registration of each source, in cases where there are source specific requirements or concerns regarding safety (for example, fractionated palm fatty acid distillates, mineral complexes (cobalt, copper, manganese, zinc etc. amino acid chelates)).
Commercial Feed Mills
- The number of mixed feed products that will require mandatory pre-market CFIA registration will be reduced.
- For feed products that will continue to require pre-market registration, the manufacturer must register the feed and provide any supporting information before the feed is sold.
- Mixed feeds must meet all of the regulatory standards that apply to their products (for example, contaminant limits, only contain authorized feed ingredients, etc.).
- Any medications in mixed feeds must be authorized for that use and used in accordance with the CMIB or a veterinary prescription.
- Feeds must be labelled in accordance with the proposed modernized labelling standards.
- Additional flexibility for allowing information on the label without requiring registration has been provided by allowing additional useful guarantees and permissible claims.
- Mixed feed manufacturers will be required to perform a hazard analysis and create and implement a written preventive control plan to address risks posed to public, animal and plant health and the environment posed by the hazards.
- Sector-specific domestic programs (for example, FeedAssure) that include compliance with feed regulatory requirements as a specification may be considered as Model Systems and be taken into consideration by CFIA in the design and delivery of risk-based inspection oversight programs.
- A permission may be required where feed manufacturers sell or distribute mixed feeds inter-provincially.
Feed Retailers/Distributors
- Where retailers/distributors package or repackage feeds received in bulk or for other identified risks, they will be required to:
- perform a hazard analysis;
- create and implement a written preventive control plan to address risks posed by the hazards to public, animal and plant health and the environment;
- and label feeds in accordance with labelling requirements.
- Feeds and feed ingredients retailed or distributed must meet all of the regulatory standards that apply to their products, including new requirements for traceability.
- A permission may be required where feeds are sold or distributed inter-provincially.
On-Farm Feed Manufacturers (Livestock Producers)
The Feeds Act generally exempts feeds made on-farm by livestock producers unless they are being sold off the farm or have a drug or other substance that presents a risk of harm to human or animal health or the environment incorporated into them.
Feeds manufactured on farm and sold:
Feeds which are manufactured on-farm and then sold, even if in small quantities, are subject to the same requirements as those for commercial feeds listed above.
- Feed manufacturers will be required to perform a hazard analysis and create and implement a written preventive control plan to address the hazards.
- A permission may be required where feeds are sold or distributed inter-provincially.
- Feeds must be labelled in accordance with the proposed labelling standards.
- If a manufacturer plans to make a feed type that will continue to require pre-market registration, they must register the feed and provide any supporting information before the feed is manufactured.
- Any medications added to feeds must be authorized for that use and used in accordance with the CMIB or a veterinary prescription.
- Feed manufacturers are responsible for ensuring their products meet all of the regulatory standards that apply to their products (for example, do not exceed contaminant limits, only contain authorized feed ingredients, etc.).
- Inspection of on-farm feed manufacturers that are selling feed will be based on the iAIM model and frequency will be dependent on a number of risk factors. Sector-specific domestic programs (for example, on-farm programs within the scope of CFIA's Food Safety Recognition Program) that include compliance with feed regulatory requirements as a specification may be considered as Model Systems and be taken into consideration by CFIA in the design and delivery of risk-based inspection oversight programs.
Feeds manufactured on farm and used on farm (i.e. not sold):
- If a feed is manufactured on farm and used on that same farm and the feed has not had incorporated into it any drug or other substance that may adversely affect human or animal health or the environment, the feed is exempt from the Feeds Act and Regulations and is not subject to CFIA oversight.
- If the feed is manufactured on farm and does contain a medication or other substance that may adversely affect human or animal health or the environment, the feed is subject to the operation of the Feeds Act and Regulations. It must meet all of the requirements as laid out above for feeds sold off the farm with some exemptions (such as for labelling requirements). Farms that manufacture medicated feed are subject to CFIA inspection as an on-farm feed manufacturer.
- Sector-specific domestic programs (for example, on-farm programs within the scope of CFIA's Food Safety Recognition Program) that include compliance with feed regulatory requirements as a specification may be considered as Model Systems and be taken into consideration by CFIA in the design and delivery of risk-based inspection oversight programs.
Importers
- Importers may be required to obtain a permission and have a preventive control plan.
- Importers are required to take responsibility for the products that they import.
- As part of the preventive control plan they will be required to demonstrate that they are ensuring feeds and feed ingredients which they import meet all of the Canadian standards including any composition, manufacturing or labelling requirements.
- Feeds must meet all of the regulatory standards (maximum limits, only use approved ingredients, etc.).
- Feeds must be labelled at time of importation in accordance with the proposed labelling standards.
- If an importer plans to import a feed type that will continue to require pre-market registration, they must ensure the feed is registered prior to import.
Exporters
- Feeds for export will be required to meet the Canadian standards for safety.
- Feeds for export will also need to meet all of the other Canadian standards unless a derogation is specifically granted by the CFIA in order to meet the importing country's requirements. Permissible derogations will be for things like labelling, or composition where safety is not compromised.
- Exporters will require a preventive control plan and may require a permission.
| Sector | Modernized Regulatory Requirements | Permissions | ||
|---|---|---|---|---|
| Preventive Control Plan | Product Labelling | Product Traceability | Licence | |
| Ingredient Manufacturer | Yes | Yes | Yes | Maybe |
| Mixed Feed Manufacturer | Yes | Yes | Yes | Maybe |
| Retailer/Distributor | Yes | Maybe | Yes | Maybe |
| Importer | Yes | Yes | Yes | Maybe |
| Exporter | Yes | Maybe | Yes | Maybe |
| Livestock Producers | ||||
| Sell off farm (medicated/non-medicated feeds) | Yes | Yes | Yes | Maybe |
| On farm use only; non medicated feeds | No | No | No | No |
| On farm use only; medicated feeds | Yes | No | No | No |
Your opinion counts
The CFIA welcomes feedback from its stakeholders and the public on the proposed regulatory changes presented in this document.
Written comments regarding any element of the proposed regulations should be forwarded to:
Animal Feed Division
Canadian Food Inspection Agency
59 Camelot Drive
Ottawa, Ontario
K1A 0Y9
Comments can also be emailed to Serge Tolusso
Comments received by April 8, 2016 will be taken into consideration prior to publication of the proposed regulations in Canada Gazette Part I
Detailed Discussion of the Proposed Regulatory Framework
Scope
The mandate of the CFIA and the Feeds Act support the protection of public health, as well as animal health, plant health, and the environment. With this in mind, the Agency proposes the following scope for a risk-based feed regulatory framework that would apply to:
- the manufacture and distribution of feed ingredients and mixed feeds ("feeds") alike, enabling oversight on an "end-to-end" approach throughout the feed supply chain.
- feeds for food-producing animals, including species not currently defined as "livestock" in the Act and Regulations (e.g. deer, elk, bison, and other mammals and birds that produce food products [meat, milk and eggs]).
- domestically produced and imported feed (approaches to the application of the framework on imported products is provided in this proposal).
- feeds manufactured for export.
- hazards with human health, animal health, plant health, and environmental implications.
The proposed regulatory scope aligns with the current scope of the Feeds Regulations, with the exception of feeds manufactured for export. New authorities in the Agricultural Growth Act expand the authority in the Feeds Act to include feeds manufactured for export. New or modernized approaches are being proposed to continue regulating safe feeds for livestock.
"Regulated parties" are those to whom the Feeds Act and Regulations are applicable. As indicated in the Feeds Act, no person shall manufacture, sell or import into Canada any feed unless the feed is registered as prescribed, conforms to the prescribed standards and is packaged and labelled as prescribed. However, the Feeds Act also indicates that it does not apply in respect of a feed that is manufactured by a livestock producer if it is not offered for sale and has not had incorporated into it any drug or other substance that may adversely affect human or animal health or the environment. This means that regulated parties are those that are involved in the manufacture, import, export and sale of feeds. However, if a feed is made on-farm by a producer as long as that feed is not sold (i.e. it's used on the farm that made it) and does not have drugs incorporated into it, the manufacture of that feed is exempt and the Feeds Act and Regulations (including the changes proposed in this document) would not apply. Should a producer make a feed and sell it, that would be considered a commercial activity and they would become a regulated party. Or if a producer makes a feed for their own use, but it includes a drug, they would also become a regulated party and need to comply with the regulations, including the changes proposed here.
Modernized Regulatory Requirements
Species
The current Feeds Act and Regulations apply to feeds for certain livestock species, where "livestock" is defined in the Act as follows:
- "Livestock" means horses, cattle, sheep, goats, swine, foxes, fish, mink, rabbits and poultry and includes such other creatures as may be designated by regulation as livestock for the purposes of this Act.
In addition, the Regulations further define:
- "Cattle" means animals of the species Bos Taurus or Bos indicus; and
- "Poultry" means chickens, turkeys, ducks and geese.
A number of other food-producing animals are not currently included in that definition. It is proposed that the scope of the species to which the Feeds Regulations apply would be expanded to include feeds for animals that are raised for human consumption. This would include:
- Game birds (e.g. quail and pheasant)
- Cervids (e.g. deer, elk)
- Bison (e.g. Bison or Bison bonasus)
- Camelids (e.g. alpaca)
- Ratites (e.g. ostrich, emu)
- Fish (clarifying which species would be included)
- Honeybees
Proposed Approach To:
Species: The scope of the species to which the Feeds Regulations apply would be expanded to include feeds for animals that are raised for human consumption. This would include:
- Game birds (i.e. quail)
- Cervids (i.e. deer, elk)
- Camelids (i.e. alpaca)
- Ratites (i.e. emu)
- Fish (i.e. non-salmonids)
- Honeybees
In addition, feed for species such as mink and fox which do not enter the food or feed chain would no longer be regulated.
Feed for species such as mink and fox which do not enter the food or feed chain would be removed from the list of species for whose feeds would be subject to the regulatory framework.
To facilitate this change, the Agricultural Growth Act amends the definition of "livestock" in the Feeds Act as follows:
- "livestock" means any animals designated by regulation as livestock for the purposes of this Act;
This definition will not change until the modernized regulations come into effect.
Expanding the scope of species would allow for more consistent application of the Feeds Regulations.
The feed that animals consume can result in residues in meat, milk and eggs if the feed risks are not controlled. By expanding the scope of species to include other farmed animals, consumers can rest assured that the feeds these animals consume are subject to regulatory controls and CFIA oversight. As mink and fox are raised for their fur, and none of their products or by-products enter the food or feed chain, it is considered appropriate for CFIA to remove the oversight of their feeds for the scope of the regulations.
Have your say
The CFIA is seeking comments on its proposal to:
- Expand the scope of species for which feeds would be subject to the regulatory framework (food-producing species of game birds, cervids, bison, camelids, ratites and honeybees);
- Clarify the species of fish for which feeds would be subject to the regulations; and
- Remove mink and fox from the scope of the livestock definition.
Standards and General Requirements
Current standards in s. 14-25 of the Feeds Regulations focus on composition and characterization of feeds (s. 14 approved ingredients, feed-additive medications; s. 20-22 composition), record-keeping (s. 15 for customer-formula and consultant-formula feeds, veterinary prescription feeds), eligibility of certain feeds for registration (s. 17-18), hazardous and other extraneous materials in feed (s. 19), specifications for guaranteed analyses required on labels (s. 24) and analytical tolerances applied to test results in relation to guarantees provided (s. 25).
The range of existing standards will be reviewed, revised, reduced or expanded as appropriate to address regulatory objectives (outcome-, risk-based and systems-based approaches) and expected modernization outcomes.
Safety Standards
To underscore the modernized regulatory framework's focus on health and safety, it is proposed that certain safety requirements are placed in regulation. This would include maximum levels for some nutrients, maximum levels for some specific contaminants, and required caution and warning statements.
Maximum Nutrient Levels
Given the proposed removal of Table 4 in Schedule I from the Regulations, it is also proposed that some nutrients which pose health and safety concerns (to animals or via residues in animal products used as human food) have maximum limits set in regulation. This may include standards for copper, zinc or iodine for some or all species whose feeds are subject to the regulations. Additional proposals and opportunities for stakeholder consultation will be provided regarding the maximum nutrient levels before they are incorporated into regulatory framework.
Maximums for Certain Contaminants
Section 19 of the current regulations sets out limits on what a feed may not contain. This includes limits on the amount of weed seeds, fluorine, mould, must or heat damage, proteins derived from certain animals, extraneous material and aflatoxins. It is proposed that these limits are updated and additional limits are added for new contaminants of concern such as heavy metals, dioxins or other mycotoxins.
Proposed Approach To:
Feed Safety Requirements: The range of existing standards will be reviewed, revised, reduced or expanded as appropriate to address current regulatory objectives (outcome-, risk-based and systems-based approaches) and expected outcomes. This includes:
- Maximum levels for some nutrients
- Maximum levels for contaminants
Hazard Identification
As indicated in the CFIA's Feed Hazard Identification / Preventive Controls – Regulatory Framework Proposal, it is proposed that all those involved throughout the feed supply chain complete a hazard identification and assessment with respect to the feed-related activities in which they are involved. The hazard identification and assessment must be carried out on incoming materials; materials sourced in-house, feed formulation, processes, and transportation by the feed or ingredient manufacturer.
Reminder:
The term "feed" refers to both mixed feeds and ingredients.
Animal feed and feed ingredients can act as a route of entry for hazards into human food and for hazards that pose a risk to animal health, plant health (e.g. pests and diseases of domestic or international quarantine significance), or the environment (e.g. invasive plant species not known to be present in a local ecosystem). To mitigate the associated risks, hazards in feeds must be identified and assessed prior to their manufacture, distribution, and feeding to animals. Multifaceted assessments are often necessary to consider both the safety to animals as the primary consumers of the feed and safety to humans as the consumers of resulting food of animal origin (e.g. meat, milk, fish, and eggs). In some cases, the risk to persons who work with and mix feed, as well as the risk to plant health or the environment also requires assessments.
To modernize the current regulatory framework regarding the identification of hazards, the Agency proposes to
- Review and update the list of prescribed deleterious substances currently contained in section 4 of the Regulations.
- Revise the list of specific hazards and standards identified for feeds, as appropriate.
- Identify specific hazards and maximum limits in feeds in guidance (build on current RG-8).
- Include known hazards and limits, where appropriate, in ingredient descriptions set out in Schedule IV.
Preventive Control Plans
It is proposed that the regulations under the Feeds Act establish minimum standards for the manufacturing and the safety of feeds for everyone the Feeds Act applies to who imports, manufactures or sells feed, regardless of the commodity or size of operation. This would be a fundamental improvement in Canada's feed safety regulatory framework. This would include requirements that address seven key elements to good manufacturing and agricultural practices:
- Products and processes;
- Sanitation, pest control, sanitizers and chemical agents;
- Hygiene and competencies;
- Equipment and conveyances to be used in an establishment;
- Physical structure and maintenance of the establishment;
- Receiving, transportation and storage; and
- Investigation and notification, complaints and recall procedures.
The proposed requirements are integral to any feed safety system. They are recognized pre-requisites for safe feed and require that industry anticipate and take the necessary measures to address the hazards that can be present in a feed, on the equipment or in the establishment and its facilities where that feed is manufactured. Each element would address specific hazards in the feed manufacturing continuum. Regulated parties would still have to comply with all the other requirements of the proposed regulations and any other applicable regulations (such as Health of Animals).
Following the identification of hazards a preventive control plan (PCP) would be required from any regulated party that manufactures or sells feed or imports or exports feeds. Farms that make their own feed and do not incorporate into it any medications or harmful materials and do not sell it would be exempt from the Feeds Act and Regulations and would not require a preventive control plan.
The requirement for a PCP would improve feed safety by requiring that these regulated parties implement a system-based approach to their operations that focusses on prevention and management systems. The steps related to the preparation of a PCP in the proposed regulations are expected to be consistent with HACCP which is the internationally recognized approach to food safety. They include the identification of:
- All hazards
- Critical control points and other control points, and related control measures that are validated by evidence
- Critical Limits and other limits
- Monitoring procedures
- Corrective action procedures
- Verification procedures
- Record keeping procedures
It is proposed that…
Control measures will need to be validated by evidence that demonstrates they effectively control identified hazards.
CFIA will provide "model systems", drawn initially from existing information that has been validated and proven effective, when properly applied.
CFIA will also provide guidance on "validations", consistent with Codex.
In preparing the PCP, which is based on HACCP principles, the regulated party would need to identify and document the potential hazards associated with their feed or processes, and demonstrate how those hazards will be controlled. Hazards may impact food safety, animal or plant health or the environment. The PCP should demonstrate how all hazards will be controlled, not just those that have a food safety implication. PCP preparation and maintenance strengthens the management of the operations by requiring the regulated party to be actively engaged in the development of feed safety systems, including proactively determining how they will monitor their operations, respond and correct deviations as they occur, and maintain ongoing compliance.
The regulated party would be required to maintain the PCP and associated records to assess its ongoing effectiveness and ensure its continuous improvement.
The PCP would need to address how specified requirements, including elements 1-7 listed in the previous section, would be met, as applicable. It would also be where regulated parties would identify the measures they have taken to comply with other relevant regulatory requirements, including those such as packaging and labelling provisions.
The PCP will be one of the key tools used by the inspector in verifying compliance with the Act and Regulations.
Many facilities already engage in these kinds of activities. The proposed requirements will strengthen the feed-safety system and focus on the system will support the preparation of safe feed products. These businesses will be well placed to comply with the proposed Hazard Identification and Preventive Control requirements.
Annex 3 contains the proposed requirements for a written PCP.
Labelling and Standards of Identity
The proposed regulations would not fundamentally change the approach to labelling and standards of identity. They would, however, provide some additional flexibility while adding elements for safety and traceability.
Labelling provisions currently exist in the Feeds Regulations. Labels play an important role in the safe and proper use of feeds. Proper labelling allows a purchaser and user of a feed to distinguish one product from another and provides information on what the feed is and how it is to be used. Risks associated with the representation and use of feed products can be mitigated by the use of labels. Products which are not labelled, or do not have the appropriate information on the label, may be unintentionally used in a manner that is not safe and causes an animal health, food safety or environmental hazard.
The intent with these proposed regulations is to improve flexibility in labelling while better aligning requirements internationally, improving traceability and mitigating safety risks.
The existing requirements of the Feeds Act and its regulations apply to all livestock feeds manufactured, imported or sold in Canada. These requirements would be maintained in the proposed regulations.
Labelling standards for feed will also remain while being modernized to improve flexibility for things such as additional guarantees.
The CFIA previously engaged stakeholders to discuss this proposal under the Feed Labelling Regulatory Framework Proposal. Feedback from that consultation has resulted in some changes which are further outlined in Annex 2.
Claims
As proposed in the Feed Ingredient Assessment and Authorization – Regulatory Framework Proposal a list of permissible claims will be developed. The permissible claims list would be a list of feed claims that can be made on feed labels without CFIA pre-market registration of the feed. The claims would be non-proprietary, any conditions of the claim would need to be met, and the claims must be truthful and verifiable.
The CFIA, Health Canada and regulated industry will work closely together to develop a "permissible claims" list as a component of a modernized approach to feed labelling.
In May 2015, CFIA outlined an approach to accommodating a permissible claims list and invited the feed industry to submit a list of claims to the CFIA to consider in order to develop a preliminary claims list for broader consultation. It was proposed to the feed industry that this list would be incorporated into the modernized regulatory framework by reference which would enable amending on a regular basis. The CFIA will prepare and distribute a separate proposal regarding an initial permissible claims list for stakeholder review and comment at a later date.
Should a feed company wish to make a proprietary claim on a label or should a claim not appear on the permissible claims list, they would still be able to have the claim assessed and approved via the CFIA's pre-market registration process.
Collective Terms
In reviewing the feedback received to the 2013 Feed Labelling Regulatory Framework Proposal, the CFIA noted that respondents raised concerns about a proposed requirement for the labelling of a full list of ingredients on all feed labels. Most respondents indicated that allowing the use of "collective terms" for certain groupings of ingredients (as suggested by the CFIA in the context of the labelling proposal) would help reduce some burden associated with the labelling of ingredients and provide some flexibility. Conversely, a couple of respondents representing individual livestock producers raised concerns that the use of collective terms would allow feed companies to not disclose specific ingredients.
Given that a detailed outline of what collective terms might be allowed and the scope of ingredients they would comprise was not elaborated in the 2013 labelling proposal, the Agency prepared the Proposal – Feed Ingredient Collective Terms on Labels for a separate consultation on this subject. The CFIA invited comments from interested parties via a preliminary consultation from early June to mid-July 2015. A second comment period was provided to interested parties between mid-December 2015 and mid-January 2016.
Following the completion of the consultation period, the CFIA prepared the "Consultation Summary – Respondent Comments and CFIA Responses" report that includes a revised set of collective feed ingredient terms that will be permitted on feed labels.
International Labels
Labels for use on feeds intended for distribution in multiple jurisdictions (e.g. U.S., EU, Canada), are often:
- Labels that contain information in languages other than English or French
- Labels that contain information that is not the same in all languages or not the same across jurisdictions
This presents a challenge for companies who must comply with Canadian requirements, while choosing an effective way of labelling their products. To provide additional flexibility for these "international labels", while still meeting the Canadian requirements:
- Information on labels, where different from Canadian regulatory requirements, shall:
- be truthful, accurate, verifiable, mathematically-consistent;
- not be incorrect or misleading;
- not contradict or conflict with Canadian requirements (e.g., maximum levels of fish flesh pigmenting agents in feeds in different jurisdictions) with respect to risks to public and animal health;
- not be seen to represent an endorsement by the CFIA of compliance with other jurisdictions' regulatory requirements;
- Canadian labelling information must appear on the label;
- additional labelling flexibilities: apply the same flexibilities that will apply to domestic labels (additional guarantees, etc.).
- Additional label information would be permitted but must not conflict with or contradict Canadian requirements, for example:
- directions or guarantees that exceed Canadian maximums would not be allowed;
- claims that do not align with Canadian requirements / policies for feed would not be allowed;
- alternative units of measurement may be used provided the results are mathematically equivalent;
- information does not need to be identical in all languages.
- Products bearing such labels would still require registration
Pre-market registration will provide for consistent application of the regulations / policies as a single point for verification of compliance. Applications for labels with information in languages other than English or French would require a certificate of translation or attestation regarding the accuracy of the translation at the time of registration to demonstrate that label information does not conflict with Canadian standards.
This approach provides level playing field for products bearing domestic and international labels, is responsive to industry / market needs and is enforceable.
Traceability and Record Keeping
While many companies have implemented voluntary traceability systems, not all companies have the basic record-keeping practices necessary for timely feed safety investigations, recalls or withdrawals. The resulting information gaps within the feed supply chain can lead to a less efficient response to a feed safety incident.
The proposed regulations would apply the international standard for traceability established by Codex Alimentarius to all feed manufacturers, licence holders and persons importing, exporting or trading inter-provincially. The Codex standard calls for tracking of feed forward to the immediate customer and backwards to the immediate supplier ("One step forward, one step back" ). These requirements are aimed at enhancing consumer protection during a feed safety incident by providing for more accurate information to facilitate the rapid identification of the origin and movement of a feed through the feed supply chain.
Currently, in some recall situations, information is provided to the CFIA but crucial time is lost deciphering company encryptions or codes, or illegible records. The proposed regulations would address this challenge by requiring that traceability information be provided electronically, upon request, in plain text and in a format that can be imported and manipulated by standard commercial software, in French or English. The information would need to be accessible in Canada.
Proposed Approach To:
Traceability: For persons importing, exporting or trading domestically, tracing of feed forward to the immediate customer and backwards to the immediate supplier ("One step forward, one step back" ) is proposed. These requirements are aimed at enhancing protections during feed contamination incidents to expedite the rapid identification of the origin and movement of feed through the supply chain.
Additional traceability requirements are also introduced via labelling requirements such as providing contact information for the manufacturer of the feed and a lot number for all feeds.
The CFIA encourages regulated parties to be proactive in enhancing their traceability and record-keeping systems beyond the proposed minimum requirements.
Have your say
The CFIA is seeking comments on:
- If the proposed approach to feed traceability will be sufficient to expedite the rapid identification of the origin and movement of feed through the supply chain?
Permissions
Introduction
As a key component of the CFIA's modernized, Integrated Agency Inspection Model (iAIM), a "permission" is defined as an official consent (e.g., permit, certificate, licence and registration) granting legal authorization to a regulated party to conduct specified activities.
The CFIA proposes to continue to use some of its longstanding authorities to grant permissions associated with feed ingredients and mixed feeds as well as to take advantage of some new authorities provided by the Agricultural Growth Act in respect of granting permissions to persons or establishments to conduct specified activities.
Reminder:
"Permission" means…
- Licences or permits
- Registrations of products, persons or establishments
- Ingredient Approvals
- Research Exemptions
or any other CFIA authorization required before an activity may be conducted.
Current Situation
The Feeds Act and Regulations have long contained authorities to require the registration of feeds (products) for domestic sale and for imported feeds. Since the early 1980's, the categories and numbers of feeds requiring mandatory registration have decreased to the point where it is estimated that some 95% of domestic feeds are exempt from this requirement. On the other hand, all mixed feeds manufactured and imported into Canada from other countries require mandatory registration prior to importation including those that would not require registration if manufactured domestically. Conversely, there have not been authorities in the Feeds Act and Regulations to require the licencing, registration or permitting of persons or facilities to conduct specified activities.
The CFIA has been issuing permits to operators of domestic inedible rendering plants in Canada since 1997 as part of its BSE prevention programming using authorities provided by the Health of Animals Act and Regulations. Among other end-uses, rendered animal by-products are commonly used in livestock feed rations as ingredients, for example, meat and bone meal, feather meal and tallow. All rendering plants must operate under a CFIA permit; conditions on permits and inspection frequencies are tied to the risks of the activities performed and products manufactured by the rendering plants.
Permissions – Ingredients
Authorization
Canadian feed manufacturers and livestock producers benefit from access to a large number of approved ingredients for use in feeds. Ingredients are assessed and approved for use in livestock feed before being added to the list of approved ingredients. As outlined previously in the Feed Ingredient Assessment and Authorization – Regulatory Framework Proposal, a number of flexibilities are proposed for ingredient approvals. It is proposed that there will continue to be a list of approved feed ingredients, and only ingredients on that list may be used to manufacture livestock feeds. As in the current framework, it is proposed that before an ingredient is imported, manufactured or sold in Canada, proponents will need to apply for authorization for any feed ingredient that:
- is new (i.e. not on a positive list);
- has been modified such that it differs significantly from a conventional ingredient; or
- is represented for a purpose other than the one(s) for which it has already been authorized.
As part of the ingredient assessment and authorization framework it is proposed that:
Application Format – as in the current regulations it is proposed that an application must be made for ingredient authorization. While applicants will have to submit all of the required information in their application package, the format and presentation of that application package will not be dictated by a rigid format. This will minimize the applicant's regulatory burden by allowing them to use or reformat an existing application package (such as one submitted to another country) rather than having to submit a package prepared specifically for Canada.
Notification of New Applications – At the present time, the CFIA does not disclose whether applications for new or modified ingredients have been received or the status of the application in the evaluation queue. It is proposed that the CFIA adopt a "notice of submission" mechanism for all feed ingredient applications that will make this information publically available
International Data – The framework will continue to require that a proponent provide satisfactory scientific evidence for the assessment and authorization of ingredients. Rather than having only one authorization pathway as is the case in the current framework, it is proposed that the type and amount of information required in support of an ingredient assessment will be subject to one of three authorization pathways:
- For a feed ingredient that is new to the global market – If Canada is the first country in which approval is sought and where it will be marketed (i.e. it has not been approved as a feed ingredient anywhere else in the world), a full assessment process and data package will be required to support the application to have the ingredient authorized.
- For a feed ingredient that is new to the Canadian market – If an ingredient is already authorized as a feed ingredient in another country and the following criteria are met, a modified application package would be allowed:
- If the ingredient has already been authorized in a country which the CFIA has recognized as having an authorization process that is equivalent to that used by Canada; a modified application package would be allowed. An applicant would need to submit evidence of the foreign authorization and a summary characterizing the ingredient and indicating it is the same ingredient, its intended purpose and directions via the application process.
- Voluntary submission of data – Proponents that have authorized ingredients which are eligible to submit a modified application package may still choose to submit the full package on a voluntary basis
In cases where a modified application is submitted for assessment, it is anticipated applicants would benefit from a shorter service standard and a reduced user fee once adjustments to operational procedures have been implemented.
If, however, the ingredient has been authorized in a country which the CFIA does not recognize as having an equivalent authorization process, a full data package will be required.
Intended Purpose Assessment – CFIA proposes to continue to require that feed ingredients have an intended purpose or purposes identified that corresponds to the definition of a feed and allows for proper categorization of the product (for example, as a protein feed, energy feed, vitamin etc.). For feed ingredients whose intended purpose would not correspond to a purpose attributed to a feed (for example, ones which would not supply a nutrient that is required by livestock), evidence to support a recognized feed purpose would be required to make the application eligible for authorization as a feed. This approach is consistent with other international feed regulatory jurisdictions where ingredients are classified and regulated on the basis of purpose and scientific evidence in support of the intended purpose is provided.
In response to stakeholder feedback regarding its role in assessing and authorizing feed ingredients, the CFIA plans to redefine its role with respect to ingredient performance assessment. Submission and pre-market assessment of scientific evidence supporting ingredient performance would only apply where failure of a product to perform an identified purpose would have significant health or safety implications to animals or humans. The scope of this approach would include the submission of evidence to demonstrate nutritional essentiality. Other examples of products for which evidence of performance would have to be submitted for assessment would include information about the bioavailability of a nutrient or the ability of an ingredient to fulfill an intended effect (e.g. prevent feed oxidation, adjust feed pH ). Categories of ingredients or criteria for which purpose evidence would not be required would be established by policy.
Safety Assessment – Feed ingredients must be safe. Consultations to date with stakeholders have confirmed they feel that the government has a central role to play in the assessment and authorization of ingredients within the scope of safety in respect of animal health, human health (via food residues and worker/by-stander exposure), and the environment.
The Regulations will be designed to continue to provide objectives and general criteria which proponents will need to consider when preparing the safety component of an application for an ingredient assessment, while still providing flexibility for applications to be tailored based on history of use and complexity of the product. The general criteria would include aspects pertaining to the identity and characterization of an ingredient (e.g. what it is, what it will be used for, how it was developed, etc.), intended purpose of the ingredient, information requirements to be used to identify risks, and manufacturing process.
Consultation – To enhance the transparency of the ingredient authorization process and meet the CFIA's requirements for consultation, it is proposed that the CFIA consult with stakeholders on the results of its ingredient assessments and recommended outcome prior to final authorization. The CFIA proposes that a post-assessment notification and consultation approach be taken prior to final authorization for ingredients which have been assessed and found to be acceptable for inclusion in the list of authorized ingredients by the CFIA. The applicant would be notified and asked to supply a description of the product and relevant information that was presented to the CFIA for assessment. This information would then be posted on the CFIA website, and interested parties notified via listserv, for a short period (e.g., 2-4 weeks) of public review and comment. The CFIA will review input from the public on the proposed decision and will consider scientific or economic rationale submitted in favour of or opposed to the decision. The CFIA will either reconsider the ingredient in light of the new information or issue a final approval of the ingredient and begin the process to add it to the list of approved feed ingredients.
Proposed Approach To:
New Ingredients: Ingredients will be assessed and approved for use in livestock feed before being added to the list of approved ingredients.
- Data from international approvals will be considered
- Ingredients must demonstrate a feed purpose
- Safety assessments will continue to be required
- New ingredients will be subject to consultation prior to final approval
A positive list of approved feed ingredients is proposed to be incorporated by reference to aid in timely updates.
Registration
Beyond the assessment and authorization of new ingredients for feeding to livestock, there are a number of ingredients that, by virtue of their being listed in Parts II of Schedules IV and V, are subject to mandatory registration of the ingredient on a source-by-source basis. This higher level of control is necessary to confirm and re-affirm (upon the expiry and renewal of registration) the consistency of the ingredient over time, the specifics of manufacturing and analytical practices employed and other risk factors.
The table below provides information on how many feed ingredients are currently authorized by CFIA for feeding to livestock in Canada and the distribution of these ingredients between the Schedules and Parts of the Schedules to the Feeds Regulations.
| Schedule # | Number (% of total) – Authorized Ingredients Part I |
Number (% of total) – Authorized Ingredients Part II |
Total |
|---|---|---|---|
| Schedule IV | 703 (84%) |
135 (16%) |
838 |
| Schedule V | 386 (99%) |
2 (1%) |
388 |
| Total | 1,089 | 137 | 1,226 |
In section 5.7 of the CFIA's Feed Ingredient Assessment and Authorization – Regulatory Framework Proposal, it was proposed that CFIA would modernize aspects of its approach to ingredient regulation by:
- Reviewing existing ingredients for alignment with their descriptions, compositional criteria, current science, intended use and safety to animals, humans and the environment;
- Making amendments to the purpose of ingredients currently in the Schedules and creating new categories or groupings of categories for ingredient types based on intended purpose, especially for the Class 8 Miscellaneous Products listed in Schedule IV; and
- Considering merging Parts I and II of the schedules into a single list.
It is further proposed that the registration of individual sources of certain ingredients, regardless of whether they are of domestic or international origin, continues in the future. The basis for ingredient registration will focus on issues of higher risk to health and safety attributable to the source of the ingredients (for example, by-products or wastes from industrial activities that may result in the ingredient having elevated levels of contaminants beyond levels known or reasonably foreseeable in other feedstuffs).
Positive List of Authorized Ingredients
Canada has benefitted from employing a positive list of authorized feed ingredients in Schedules IV and V, and this use of a positive list approach continues to be supported by stakeholders in consultations to date. As shown above, the current list already contains over 1200 ingredients that are categorized according to intended purpose, are described, and identify appropriate guarantees as necessary. Only those ingredients, listed in these schedules, may be used in the manufacture and sale of feeds in Canada, or in feeds imported into Canada.
In section 6.4 of the CFIA's Feed Ingredient Assessment and Authorization – Regulatory Framework Proposal, it was noted that timely amendments to the ingredient Schedules that form part of the Regulations has been problematic. As an alternative approach, the CFIA had an interest in pursuing the ability to incorporate, by reference, documents into regulations that are externally or internally generated, and that may change over time. This would provide increased flexibility and timely updating.
In February 2015, section 5.1 of the Feeds Act, by way of the Agricultural Growth Act, came into effect where:
5.1 (1) A regulation made under subsection 5(1) may incorporate by reference any document, regardless of its source, either as it exists on a particular date or as it is amended from time to time.
Use of this new incorporation by reference authority will be explored as a mechanism to enable having a discrete, enforceable positive list of authorized feed ingredients in the modernized feed regulatory framework so that Schedules IV and V can be removed from the structured regulation and be referred to as a list that the CFIA maintains outside of regulation. This will allow for more timely updates to the list of ingredients so that industry and inspection staffs have transparent access to the most current list of ingredients. Part of this includes the need to provide adequate consultation, as proposed above under the heading "Consultation" above.
In September 2014, CFIA signalled to its stakeholders that a comprehensive review of currently-authorized feed ingredients was being initiated and industry experts were invited to express their interest in collaborating with CFIA on the review of groupings of ingredients (for example, oilseed products, vitamins etc.). As part of this review project, the scope includes determining whether certain ingredients which are currently required to be registered by source should continue to be registered and if there are others that are not subject to registration at present but should be going forward. Once the range of authorized ingredients has been reviewed and compiled, the CFIA will propose a revised list with the updated ingredient descriptions, including any changes to ingredients subject to registration, and seek stakeholder feedback on the proposal. Given the scope of this project, it is not expected to be completed by the time the new regulations come into effect. Any changes that have been completed will be included and the CFIA will continue to work collaboratively with industry to update and modernize the list in the future.
Additional analysis may be needed to verify that the authorized ingredients are also suitable for the additional species that are proposed to be brought under the definition of livestock.
A modernized and relevant list of authorized feed ingredients will continue to allow feed manufacturers to take advantage of a wide range of products when formulating livestock feeds.
Permissions – Mixed Feeds
Registration
Pre-market assessment and registration of mixed feeds is currently one of the regulatory mechanisms used to help ensure the safety and efficacy of livestock feeds sold in Canada. The regulations currently exempt the vast majority of standard feeds, such as complete feeds, supplements, customer formulas and veterinary prescription feeds. These are estimated to account for approximately 90 – 95% of the feeds manufactured in Canada. The remaining types of feeds, which require registration, typically represent feeds that are not as straight forward such as feeds that may have higher concentrations of nutrients, directions for use that are not standardized, feeds that are more variable due to source differences, or those feed types that are less typical (such as enzyme supplements). In addition, feeds that have a claim on the label or which have additional label information (such as extra nutrient guarantees) also require pre-market registration.
Historically, all feeds required pre-market registration. When the regulations were last updated in the early 1980's, a number of exemptions to registration were granted for types of feeds that were well understood at the time. As the regulatory modernization process is moving forward, it has been recognized that past knowledge and familiarity with some feed types has grown sufficiently that the CFIA is taking this opportunity to examine the importance pre-market registration plays for other feeds.
The requirement for pre-market registration is viewed as a costly and unnecessary step by some regulated parties. In addition, due to the volume of submissions received, it can take a few to many months for a new feed to be registered, thus delaying the entry of that product into the Canadian market. Currently, both registered and unregistered feeds are subject to CFIA inspection oversight during routine feed mill and retail inspections.
It is proposed that additional types of feeds be exempted from registration. Given changes in technology and knowledge of nutritional requirements for livestock, pre-market registration does not continue to play a useful role for all feed types. However, there are a few categories of feeds that, at a minimum, will continue to require registration as there are health and safety or consumer protection concerns associated with these feeds, including:
- Milk replacers
- These are fed to young animals as a sole source of nutrition. Failure to provide adequate nutrition or provide clear and appropriate feeding instructions can quickly result in issues of animal health.
- Feeds delivered via water
- A number of vitamins and other supplements are being fed to animals via their water. Since water intakes are highly variable and dependant on activity, external temperature and even feed composition (such as salt intake), there is concern that feed supplements that are delivered through the water can be consumed at unsafe levels which may result in issues of animal health or residues in the animal products.
- Medicated Minerals
- These minerals are not typically mixed into a complete feed and there are concerns regarding intake of the mineral and thus the medication may not be delivered at the correct level. Excessive consumption may result in animal health or food safety concerns (residues) and under medication results in medications not working effectively. Oversight of these by way of product registration helps to address exposure concerns.
- Flavouring agents
- These are mixtures of flavouring ingredients. All flavouring ingredients have maximum use rates for safety. Combinations of these individual flavouring ingredients can result in their use at levels that are higher than approved. In addition, the use of the flavouring agent needs to be set at levels that do not present a safety risk.
Table 4 (Schedule I)
Table 4 of Schedule I sets out nutritional standards which are used to exempt complete feeds and some supplements from registration. Currently, if a complete feed provides nutrients which fall within Table 4 or a supplement has directions for use which would result in a complete feed that provides nutrients which fall within Table 4 the feed can be exempted from registration. Both CFIA and stakeholders recognize that the values in Table 4 no longer have the same nutritional relevancy that they did when the table was introduced. In addition, stakeholders have indicated that they feel Table 4 prevents innovation for new feed products. Feeds will continue to be required to be suitable for their intended purpose and must meet an animal's nutritional requirements. As some of the maximum limits which are currently set in Table 4 have health and safety implications for animals and for residues in animal products, it is also proposed that maximum limits for certain nutrients be set. See the section on Safety Standards above for further details on maximum limits in feeds.
The CFIA will prepare and distribute separate proposals for stakeholder review and feedback regarding nutrient maximums by species or classes of species (for example, for broiler versus laying chickens) as appropriate as part of its on-going regulatory development and consultation activities.
Customer and Consultant Formula Feeds
Customer and consultant formula feeds are defined types of mixed feeds in the Regulations which are currently exempted from registration. Given the proposed flexibilities outlined above, it is also proposed that customer and consultant formula feed types will be removed from the regulations. Customer formula feeds were designed to allow a producer to request that a manufacturer make a specific formula for them without having to comply with the exemption from registration criteria. Given that Table 4 nutrient restrictions will be removed and most feeds will be exempt from registration, the customer formula exemption would no longer be necessary. Feeds that are proposed to continue to require registration are due to health and safety concerns, and any maximum limits set will also be due to health and safety concerns so all feeds, whether requested by a producer or made commercially by a manufacturer should meet those requirements. Consultant formula feeds were designed to exempt feeds that are formulated for a specific purchaser.
Given the flexibilities being introduced to further reduce registration requirements and that any restrictions would be based on health and safety concerns, it is proposed that the:
- "customer formula feed" definition will be retained but modified to accommodate on-farm feed manufacturing for own use and enable the application or exemption of certain regulatory requirements (for example, concerning the manufacture of medicated feeds) not exempted from the scope of the Feeds Act; and
- "consultant formula feed" definition and exemptions will no longer be required.
Have your say
The CFIA is seeking comments on its proposal to:
- Retain a modified "customer formula feed" definition and requirements focussed on on-farm feed manufacturing not exempted from the scope of the Feeds Act; and
- Remove the "consultant formula feed" definition and exemptions from the Regulations.
Veterinary Prescription and Medicated Feeds
It is proposed that there is no change to the approach for veterinary prescription and medicated feeds. The addition of a medication, either via a veterinary prescription or as per the CMIB, is currently allowed without the medication being a trigger for registration. It is proposed that this continues in the new regulations. The feed to which the medication is being added must be compliant with the regulations and meet any standards or labelling requirements.
Stakeholders are advised that as a response to the Federal Action Plan on Antimicrobial Resistance and Use in Canada, Health Canada's (HC) Veterinary Drugs Directorate (VDD) is preparing several initiatives including measures to increase the oversight of medically-important antibiotics used in livestock feeds. As CFIA partners with HC to regulate medicated livestock feed in Canada, amendments to the Feeds Regulations may be necessary to accommodate proposed HC policy and regulatory changes. It is anticipated that HC will lead a separate stakeholder consultation, with CFIA support, regarding proposed policy and regulatory change.
Permissions – Persons or Establishments
Domestic (Inter-provincial) Trade
Individual and establishment permissions represent a tool to identify a regulated party and authorize him, her or the establishment to conduct an activity. The CFIA proposes to grant permissions authorizing regulated parties to conduct specific activities to meet regulatory requirements.
Two types of permission are envisioned:
- Permissions with a Preventive Control Plan (PCP) as a condition (where parties with frequent transactions will be required to have effective PCPs in place in order to be granted a permission); and
- Permissions without a Preventive Control Plan (where parties participating infrequently in regulated activities subject to regulatory requirements (e.g., one-time imports or exports of a regulated article) may be eligible for permissions without PCPs.
The condition that certain permission holders develop, implement and maintain effective preventive control plans (PCPs) suitable to the size and complexity of their operations is a key requirement of the permissions regime. This is integral to regulated parties assuming greater responsibility for the safety and compliance of their products. Details on the proposed elements of a feed preventive control plan and the expected outcomes and performance criteria are outlined in the CFIA's Feed Hazard Identification/Preventive Controls – Regulatory Framework Proposal.
The CFIA would place a higher priority on oversight of regulated parties by way of permissions where the consequence of non-compliance would have a greater impact on human health, animal health, plant health or the environment. Regulated parties who strive to adopt the best practices and innovative, commercial or technological strategies would be recognized by the CFIA through adjustments to oversight, as appropriate.
Permissions would describe all relevant activities the applicant is approved to conduct. An application may be made for each physical location, a number of physical locations, or by activity (for example, import, export), at the discretion of the applicant.
The following sections of this consolidated proposal will discuss these two types of permissions in greater detail.
Legal Authorities
Recent amendments to the Feeds Act by way of Agricultural Growth Act provide expanded authorities to the CFIA to issue permissions with respect to operators and establishments involved in feed-related activities.
5.2 (1) The Minister may, on application, register a person, or issue a licence to a person, authorizing them to conduct a prescribed activity in respect of a prescribed feed that has been imported for sale—or that is to be exported or to be sent or conveyed from one province to another—or both register a person and issue them a licence.
5.3 (1) The Minister may, on application, register an establishment as one where a prescribed activity may be conducted in respect of a prescribed feed that has been imported for sale or that is to be exported or to be sent or conveyed from one province to another.
5.4 Subject to the regulations, the Minister may amend, suspend, can cel or renew a registration made under subsection 5.2(1) or 5.3(1) or a licence issued under subsection 5.2(1).
The Agricultural Growth Act has also been used to amend a broad exemption provision in the Feeds Act that applies to livestock producers who manufacture feeds at the farm level:
4. This Act does not apply in respect of a feed that is manufactured by a livestock producer if it is not sold and has not had incorporated into it any drug or other substance that presents a risk of harm to human or animal health or the environment.
Scope
With the new authorities from the Agricultural Growth Act, under the proposed regulations, domestic manufacturing and distribution of feed constituting trade in an inter-provincial context could be required to obtain a permission from the CFIA and comply with the requirements of the Feeds Act and its Regulations or other conditions set out in the permission.
As context for prescribing activities and prescribing feeds in regulations as required by sections 5.2(1) and 5.3(1) of the Feeds Act set out above, the following factors could be taken into consideration for establishments in Canada:
- product type (e.g. ingredients, mixed feeds, medicated feeds);
- intended use (e.g. for sale directly to customer, for resale);
- intended species (e.g. ruminants, poultry, multiple species);
- processing activities (e.g. pelleting, extruding, drying);
- annual volume of production; and
- use of higher risk materials (e.g. medications, prohibited material).
The requirement for a permission could further be restricted to instances where risks posed by hazards are considered as known or reasonably foreseeable from activities and/or in feeds and feed ingredients. Hazards would include something inherent in a feed that can pose a risk of harm.
Bearing these factors in mind, it is proposed that the following criteria will be applied in the prescribing of activities and of feeds where operators or establishments would be subject to requiring permissions with Preventive Control Plans as a condition:
- Manufacture, package, label, sell any feeds which are subject to warnings (public health risks) and cautions (animal health risks) with respect to ingredients and mixed feeds
- this could include feeds for non-ruminants containing prohibited animal proteins, medicated feeds, certain ingredients or feeds containing certain ingredients where cautions and warnings will be required to be labelled and carried forward onto labels of other feeds containing these products;
- certain medicated feeds that are not required to be withdrawn prior to animals being shipped for processing and pose minimal risks to non-target species could be exempted
- Manufacture, package, label, sell, any feeds which are subject to any maximum use restrictions (via ingredients or other feeds containing such ingredients)
- Example – carotenoid pigments added to feeds to colour flesh in fish;
- Establishments not dedicated to the manufacture of feeds or feed ingredients to require permission
- Example – facility makes livestock feed and pet food in same facility;
- Example – facility make biofuel as principal product; distillers' dried grains (DDGs) are co/ by-products from biofuel production for sale or distribution into the livestock feed supply chain.
Depending on the criteria that would be applied to the requirement for a permission, issuance of permissions in light of "emergency" circumstances may also be necessary to address temporary interruptions in the supply of feeds, for example
- a feed manufacturer in one province steps in to supply customers of a feed manufacturer in a neighbouring province which has suspended operations due to a fire or to address other urgent operational issues.
In the case of permissions that would not have PCPs as a condition, it is proposed that they would be required in the following instances:
- Human food manufacturers already complying with a preventive control plan under the SFCR would not need a separate plan when supplying a by-product of food manufacture to feed, provided the feed would still be suitable as a food and is not further processed.
The proposed permissions would be valid for a period of two years and subject to a fee to be identified in a user fee proposal being developed for consultation at this time. Regulated parties would be able to apply for multiple permissions (for each establishment, activity or commodity) or a single permission (for all their establishments, activities and commodities).
The permission application would serve to collect basic information about the prospective permission holder, where feed businesses are located and key activities (e.g. manufacturing feed to be exported or traded inter-provincially, importing and exporting with a certificate).
Exemptions
It is further proposed that regulated parties not required to obtain a permission could include the following:
- primary producers, such as livestock producers or their operations, are not expected to be covered by the proposed domestic permissions regime provided they do not sell or convey feeds across provincial boundaries;
- feed or feed ingredient manufacturers who are not involved in trade across provincial boundaries;
- transporters of feed, unless there is a plant or animal health requirement to address potential risks of transmission of diseases or pests posed by the movement of feeds between provinces; and
- facilities that store or retail feeds but are not involved in importing, exporting or preparing the commodity, provided it remains packaged and sealed from receipt to transportation, and meets the requirements of the relevant legislation.
Anticipated Outcomes
The issuance of permissions in this regard would improve the existing regulatory environment by:
- providing an accurate, current and complete picture of who is involved in the preparation of feed in inter-provincial trade in Canada allowing for the collection of essential data to conduct oversight on the basis of risk;
- enabling industry to take a more systematic approach to PCPs to cover activities in multiples locations or activities across multiple programs (such as feed and food);
- providing an additional enforcement tool that can be acted upon to compel compliance/respond to non-compliance in situations where risks of harm to public, animal or plant health or the environment from feeds are identified;
- enabling better CFIA oversight of higher risk prescribed activities and feeds
- triggering of notification by permission holder and inspection by CFIA where holder seeks to move to activities/feeds of a higher level of risk than their existing permission authorizes them for;
- having a single, consistent approach to requirements for permissions across CFIA programs; and
- implementing a scheme based on a systems approach.
Have your say
The CFIA is seeking comments on:
- Are there other situations where it would be appropriate to require a permission that would not have PCPs as a condition?
- Are there other situations or businesses that would want to choose to obtain a permission even if they are not obligated to have one? Would allowing "voluntary licensing" be beneficial to industry?
Given that:
- the iAIM proposes to exempt transporters from the requirement to obtain a permission unless there is an animal or plant health requirement; and
- section 170(1) of the Health of Animal Regulations obliges persons operating a conveyance to have procedures in place to prevent the contamination of animal food for ruminants with prohibited material (animal health risk). Further, there are other hazards that could pose a food safety and/or animal health risk if not properly managed during the transportation of bulk feeds. This is especially the case where conveyances are used to transport a range of feeds and non-feed items.
- Are there specific situations related to transportation of feeds where a requirement for permission for feed transporters might be necessary to control risks?
Importers
Canada is dependent on imports of feeds and feed ingredients to enable the manufacture of highly cost-effective and nutritionally-effective feeds that contribute significantly to competitive livestock production systems. Over $269 million of animal feed products were imported into Canada in 2013. The main countries of origin include feed imports from the United-States (78.68%), France (7.3%) and China (3.72%).
To protect the health of the animal and plant resource bases and the environment, imports must be free from pests and diseases. To support a fair and competitive marketplace, products entering Canada must also meet domestic feed regulatory requirements, including those for safety, nutrition, composition, product identification (e.g. labelling), packaging and quality. With the ever-increasing rate of global trade comes ever-increasing risk to our domestic animals and plants. As the number of countries exporting feeds to Canada increases, the robust oversight of feed imports is a key component of the modernized regulatory framework.
Current Situation
With respect to the importation of feed ingredients, the current regulatory framework takes a two-tiered approach to oversight:
- For ingredients that the Agency has assessed, authorized and listed in Parts I of Schedules IV and V to the Feeds Regulations (representing many common grains, oilseeds, rendered animal proteins and fats, minerals, flavouring agents), oversight is provided by the Canada Border Services Agency (CBSA) and the CFIA at the border. Documentation for imports is checked for conformance (including any applicable to animal and plant health) with regulatory import requirements set out in the Automated Import Reference System (AIRS). The vast majority of authorized ingredients are contained in Parts I of both Schedules (84% and 99% respectively) subject to this level of oversight.;
- For ingredients that the Agency has assessed, authorized and listed in Parts II of Schedules IV and V to the Feeds Regulations, CFIA registration of each individual source of the listed ingredients is required prior to the ingredient being allowed entry into Canada. Registration of ingredients in Part II serves to enable a closer scrutiny of safety or other risks that are ingredient-specific. Confirmation of registration and conformance with other requirements as set out in AIRS is also undertaken by CBSA and CFIA at the border prior to entry.
- Please refer to the "Ingredient Authorization" section of this proposal above for a more detailed discussion about anticipated changes to feed ingredient assessment and authorization by the Agency going forward.
With respect to mixed feeds,
- All mixed feeds originating from outside of Canada require mandatory CFIA registration prior to importation. Registration enables the CFIA to confirm compliance with domestic regulatory requirements prior to the products being allowed entry into Canada.
- Imported mixed feeds are subject to the same registration fees that apply domestic feeds as set out in chapter 1 (Table 1) of the Agency's RG-1 Regulatory Guidance: Feed Registration Procedures and Labelling Standards.
The table below provides information on how many feed ingredients and mixed feeds are registered by CFIA for sale in or importation into Canada and the distribution of these registrations between Canadian and foreign registrants.
| Feed Type | Number (% of total) – Feed Product Registrations Canadian |
Number (% of total) – Feed Product Registrations Foreign |
Total |
|---|---|---|---|
| Feed Ingredients (% of total) |
264 (51%) |
259 (49%) |
523 |
| Mixed Feeds (% of total) |
3,703 (71%) |
1,484 (29%) |
5,187 |
| Total (% of total) |
3,967 (69%) |
1,743 (31%) |
5,710 |
The table below provides a summary of the top 10 countries outside of Canada with registrants for feeds registered by the CFIA.
| Country | Feed Ingredient Registrations (#) |
Mixed Feed Registrations (#) |
Total Registrations |
|---|---|---|---|
| USA | 226 | 1,379 | 1,605 |
| France | 4 | 40 | 44 |
| United Kingdom | 2 | 20 | 22 |
| Belgium | 1 | 14 | 15 |
| Scotland | 0 | 8 | 8 |
| Denmark | 2 | 6 | 8 |
| Spain | 0 | 7 | 7 |
| Netherlands | 5 | 1 | 6 |
| Switzerland | 1 | 4 | 5 |
| Germany | 2 | 3 | 5 |
| All Other (9) Table Note 1 | 13 | 5 | 18 |
| Total | 259 | 1,484 | 1,743 |
Table Note
- Table Note 1
-
includes Brazil, Finland, Malaysia, Japan, Norway, Italy, Ireland, Trinidad and Tobago, and India
Post-entry compliance of the actual imported feeds themselves is most often verified at destination (feed retail and commercial feed mills), but these may not be the most appropriate sites to have any non-compliance corrected.
With respect to domestic traceability of feed imports, many importers of rendering plant products (for example, meat and bone meal, feather meal, blood meal, poultry fat and other animal protein and fat products authorized for feeding to animals) and many animal foods have been subject to traceability requirements in Part XIV of the Health of Animals Regulations since the late 1990's. These requirements do not, however, extend to all imported feeds and feed ingredients. Even if some importers have implemented voluntary traceability systems for feeds not subject to the Health of Animals Regulations, not all importers have the basic record-keeping practices necessary for timely feed safety investigations, withdrawals or recalls.
Proposal – Introduction
Changes in global demand, production and distribution environments, technological advancements, coupled with industry consolidation, have resulted in significant increases in the complexity, speed and volume of trade.
In this operating environment, the CFIA should be able to identify regulated parties and understand the nature and risk of their regulated activities. Furthermore, the level of CFIA's oversight should correspond to the regulated party's ability to meet regulatory requirements.
Given the geographical size of Canada, the extensive range of air, land and sea points of entry, the distribution of domestic livestock populations and trading patterns for feed, an expansion of inspection capacity to more closely scrutinize imports of feeds at points of entry would represent a more resource intensive approach to oversight that would see CBSA and CFIA retaining the majority of responsibility for the verification of compliance of imports with domestic requirements. Such an approach runs contrary to CFIA objectives and strategies regarding responsibilities for assuring compliance and the provision of oversight. In addition, this approach would not represent a proactive, pre-entry means of addressing potential safety or other risks associated with products originating from outside Canada.
Proposal – Options for Consideration
In the case of feed imports, the CFIA is considering a range of oversight options that could offer a combination of alternative, effective and flexible approaches to enable market access while providing sufficient oversight to minimize domestic health and market risks. These options include:
1. Continuing with some manner of product permissions (ingredient and mixed feed registration) for import control (modified status quo)
Beyond the proposed mandatory registration requirements for certain feed ingredients and mixed feeds, regardless of their country of origin (as discussed above in the "Permissions – Ingredients" and "Permissions – Mixed Feeds" sections respectively of this proposal), the CFIA could review the current mandatory registration requirements imposed on all mixed feed imports using more current risk-based criteria to better identify the feeds that may pose the highest risks to public, animal and plant health and the environment and adjust the regulatory framework accordingly. Such criteria could include:
- feed type (e.g. ingredients, mixed feeds, medicated feeds);
- intended use (e.g. for import directly by end user, import for sale/distribution to end users );
- intended species (e.g. ruminants, poultry, multiple species);
- manufacturing processes (e.g. pelleting, extruding, drying);
- volumes of product imported;
- use of higher risk materials in the feed (e.g. medications, proteins prohibited from feeding to ruminants);
- compliance history; and
- typical country(ies) of origin of products.
While pre-market product assessments and permissions are often considered to be burdensome and time-consuming by industry stakeholders, there may be merit in providing for an import-specific product-by-product assessment and permissions option.
This option:
- would have CFIA retaining responsibility for verifying that the imported feed complies with domestic regulatory requirements;
- includes having the CFIA assess the adequacy of the hazard identification undertaken by the applicant for the feed to be registered and the prevent control plan implemented to address risks posed by any identified hazards;
- may not provide for a level marketplace playing field where similar products from domestic sources not subject to mandatory registration have less impeded access to the Canadian marketplace;
- could represent a potential accommodation for "voluntary" product registrations for situations where prospective feed importers or exporters would prefer this control measure and its associated compliance obligations to other control measures and their associated compliance obligations given the nature of the product, market share and other operational considerations.
To address existing traceability gaps for imports in the context of the domestic feed supply chain and as discussed in the Traceability and Record Keeping section of this proposal, this option would see importers being responsible for complying with traceability requirements based on the international standard for traceability established by the Codex Alimentarius to all feed importers.
Requiring compliance with traceability requirements would:
- improve the importer's and CFIA's ability to manage issues when a risk is identified in an imported feed;
- not represent a single measure sufficient to proactively address potential safety or other risks associated with products originating from outside Canada.
2. Requiring importers to obtain a permission with a preventive control plan (PCP) as a condition
As discussed in the Permissions – Domestic (Inter-provincial) Trade), section of this proposal, this option aligns with the guiding principles of CFIA's iAIM and approach being taken by other modernized CFIA regulatory frameworks (for example, the Safe Food for Canadians Regulations). It provides the tools to improve regulatory oversight at appropriate points in the feed supply chain.
As discussed in section 4.4 of the Feed Hazard Identification/Preventive Controls – Regulatory Framework Proposal published by the CFIA in June, 2014, importers could be called upon to demonstrate that foreign manufacturers have designed, developed, implemented, and verified preventive control plans that are comparable with those required by domestic manufacturers to assure the safety of feed and feed ingredients and compliance with other Canadian regulatory requirements.
Importers would have to have PCPs of their own that include elements which apply to their operation. Importers without facilities, for example, would not be expected to address physical structure and maintenance. However, all importers would, at a minimum, need to include the following elements in their PCPs:
- Element 1: Process and Product Controls
- Element 7: Traceability, recall, and complaints
Importers who are involved in further handling or repackaging of feeds would address all seven elements required in a PCP.
Several regulated parties currently import feed commodities into Canada to be subsequently brought into compliance before being offered for sale. This practice is particularly used to correct non-compliance related to product labelling and often on products for smaller market segments. In order to allow existing practices to continue, the scope of this option would also permit non-compliant feed to be imported or traded inter-provincially on the condition that the feed is clearly labelled with, "For further manufacture only", that it is imported to a single location and that it is brought into compliance within three months after the day on which it is imported or traded inter-provincially (unless a longer period is specified by the Minister). However, a prohibition is proposed against the mixture/dilution of imported contaminated feed with non-contaminated feed in order to bring the product into compliance. This is in keeping with international practice in the feed sector and current regulations.
This option would:
- make importers most responsible for assuring that feeds comply with Canadian regulatory requirements for safety, composition, labelling and others;
- provide an accurate, current and complete picture of who is involved in the preparation of feed in inter-provincial trade in Canada;
- enable industry to take a more systematic approach to PCPs to cover activities in multiple locations or activities across multiple programs (such as feed and food)
- increase the scope of regulated parties to which this more robust oversight approach would apply, particularly those who are importers of ingredients listed in Parts I of Schedules IV and V, where the bulk of compliance verification takes place at points of entry by CBSA and CFIA
- Consequently, this option may be viewed as intrusive and may create a disincentive for certain current importers to continue importing feeds in future.
- allow for the collection of essential data to conduct oversight on the basis of risk;
- enable better CFIA oversight of higher risk prescribed activities and feeds
- triggering of notification by permission holder and inspection by CFIA where holder seeks to move to activities/feeds of a higher level of risk than their existing permission authorizes allows;
- provide an additional enforcement tool that can be acted upon to compel compliance/respond to non-compliance in situations where risks of harm to public, animal or plant health or the environment from feeds are identified;
- represent a single, consistent approach to requirements for permissions across CFIA programs and a scheme based on a systems approach.
3. Authorizing operators or establishments in other countries (by way of permissions) to export feeds directly to Canadian customers
Under this option, feeds manufactured in other countries could be eligible to obtain a permission from the CFIA to export feeds to Canada. This option would be contingent, however, on the foreign country having been assessed and found to have comparable regulatory and oversight systems for feed as those in Canada.
This option would:
- compel the foreign exporter, by way of conditions on the permission, to assume responsibility for the compliance of products intended for the Canadian market to meet domestic regulatory requirements;
- preclude Canadian importers from having to have preventive control plans for products imported from suppliers in such countries;
- oblige importers to comply with traceability requirements in the event risks are identified in products they import; and
- enable the CFIA to modify, suspend or cancel the foreign establishment's permission in the event feeds are determined to not comply with Canadian safety or other domestic requirements.
At present, the Government of Canada has not assessed and identified any countries whose feed regulatory system is comparable to ours nor is it anticipated to be an option that will be available to importers in the immediate (1-3 years) future.
Permissions not requiring a preventive control plan (PCP) as a condition
The proposed regulations would include certain exemptions to the PCP condition on permissions for the importation of feeds, including:
- feed carried on any conveyance intended for the animal that it accompanies; or
- samples for scientific analysis, trade shows or other marketing purposes.
Transshipment of feed through Canada is also proposed for exemption from a PCP requirement, provided the shipment travels through Canada in bond.
Have your say
The CFIA is seeking comments on:
- which option or combination of options discussed above should the CFIA consider enabling in a modernized feed regulatory framework? Please include a rationale with your comments; and
- if there are any other options the CFIA should consider enabling? Please include a description of the option(s) with your comments.
Exporters
While the trade of feed in Canada is largely domestic in nature, over $381 million of Canadian animal feed products were exported abroad in 2013. The main destinations for Canadian feed exports are the United States (61.9%), Singapore (6.73%), Philippines (5.6%), Mexico (4.53%) and Japan (3.95%).
Current Situation
Currently, feeds intended for export from Canada and so labelled are exempted from the operation of the Feeds Act and the Regulations. Where trading partners seek certification by a "competent authority" as a condition of export market access, export certificates (e.g. for U.S.) and certificates of free sale are issued by CFIA on a voluntary basis.
In not regulating exports, CFIA has limited capability to prevent the export of unsafe feeds to other countries given current exemptions from the Act and Regulations
Proposal
To increase assurance that feeds in international trade originating from Canada are safe, it is proposed that all feeds manufactured in Canada intended for export meet Canadian standards for safety and other domestic compliance requirements. In keeping with the principles of iAIM and as discussed in terms of domestic trade and imports of feed, exporters would also be obliged to obtain a permission from the CFIA with a condition of having a preventive control plan to address risks posed by any identified hazards in their feeds and achieve any other domestic compliance requirements that would apply. Recent amendments to the Feeds Act by way of the Agricultural Growth Act now provide the CFIA with the authorities to:
- require exporters to obtain a permission in this regard (Feeds Act, section 5.2); and
- issue any certificate or other document setting out any information that is necessary to facilitate the export of any feed (Feeds Act, section 5.5).
Exemptions from some domestic requirements for feeds intended for export would be identified so feeds could meet a foreign country's non-health and safety compliance requirements where they differ from Canadian ones.
In the case of the export of feed ingredients not authorized by CFIA for feeding in the domestic market, exporters might still be able to obtain a CFIA export permission provided that the ingredient:
- is prepared or manufactured in accordance with Canadian regulatory requirements (for example, in accordance with an appropriate preventive control plan);
- meets any other applicable requirements specified by the importing country; and
- can be certified by the CFIA for an export market if required by the importing country.
This modernized approach to feed export oversight would:
- establish a more consistent, robust framework to support export market access opportunities;
- demonstrate clear authority and provide mechanisms of oversight for feed exports to international markets;
- align favourably with approaches being proposed for food exports under the CFIA's proposed Safe Food for Canadians Regulations and the Integrated Agency Inspection Model (iAIM); and
- enable the CFIA to modify, suspend or cancel the foreign establishment's permission in the event feeds are determined to not comply with Canadian safety or other domestic requirements.
Have your say
The CFIA is seeking comments on its proposal to:
- require exporters to obtain a permission from the CFIA to export feed to international markets;
- require exporters to have preventive control plans as a condition for obtaining permissions to export animal feed from Canada; and issue export certificates for animal feed when required by trading partners.
Permission Requirements
With new authority from the Agricultural Growth Act, under the proposed regulations, everyone importing, manufacturing feed for export or for inter-provincial trade, or exporting a feed for which they are requesting an export certificate, would be required to have a permission and comply with the requirements of the Feeds Act and its Regulations. This would improve the existing system by:
- Implementing a permission scheme that will be based on a systems approach;
- Providing an accurate and complete picture of who is involved in the preparation of feed for inter-provincial trade or export (in some cases) and import of feed into Canada;
- Allowing for the collection of essential data to conduct oversight on the basis of risk;
- Having a single, consistent approach to requirements for licensing and registration across CFIA programs.
In addition, industry would be able to take a more systemic approach to permissions and PCPs to cover activities in multiples locations or activities across multiple programs (such as feed and food).
The proposed permission would be valid for a period of two years, for a fee of approximately $250Footnote 2, and regulated parties would be able to apply for multiple permissions (for each establishment, activity or commodity) or a single licence (for all their establishments, activities and commodities).
The permission application would collect basic information about the licence holder, where feed businesses are located and key activities (e.g. manufacturing feed to be exported or traded inter-provincially, importing and exporting with a certificate). Permission numbers would be generated, as well as a number for each establishment covered by the permission. It may also collect information to enable risk-based oversight such as:
For establishments in Canada:
- product type (e.g. ingredients, mixed feeds, medicated feeds)
- intended use (e.g. for sale directly to customer, for resale)
- intended species (e.g. ruminants, poultry, multiple species)
- processing (e.g. pelleting, extruding, drying)
- annual volume of production
- use of higher risk materials (e.g. medications, prohibited materials)
- compliance history
- the number of employees and annual gross sales to identify small and micro-businesses.
For importers:
- product type (e.g. ingredients, mixed feeds, medicated feeds)
- intended use (e.g. for sale directly to customer, for resale)
- intended species (e.g. ruminants, poultry, multiple species)
- processing (e.g. pelleting, extruding, drying)
- annual volume of product imported
- use of higher risk materials (e.g. medications, prohibited materials)
- typical country of origin of products
Permission renewal every two years would ensure CFIA has up-to-date information on permission holders and their activities. This information is necessary for risk-based oversight.
Based on the information received in the permission application, an inspection prior to the issuance, renewal, or amendment of a permission may be required depending on product risk and compliance history. This is likely to be the case initially for feed that has historically been recognized as having higher safety risks, such as medicated feeds, or feeds containing prohibited materials.
When there is non-compliance, the proposed regulations would allow the Minister to suspend a permission. For example, a permission would be suspended immediately, upon notice, where there is a risk of injury to human health. This enforcement tool would complement other compliance and enforcement measures available to inspectors.
Risk-based Oversight
Introduction
Not only is feed regulatory modernization being undertaken to address objectives such as reducing regulatory burden, increasing responsiveness to industry changes and providing clarity and flexibility to affected regulated parties, it is also being shaped by a concurrent initiative to modernize the Agency's approaches to enforcement via oversight strategies and activities.
The Integrated Agency Inspection Model (iAIM) features a more prevention-focussed and systems-based approach that enables both the CFIA and regulated parties to more readily adapt to emerging global and scientific trends.
The model is predicated on having some common regulatory tools in place (such as requirements for preventive control plans and grant permissions) to enable comprehensive and consistent application across all commodities/programs. Consequently, such common tools as preventive control plans and permissions are featured prominently in this modernized feed regulatory framework proposal.
Inspection Modernization – Guiding Principles
The Agency's modernized inspection model is to apply globally recognized risk management concepts based on prevention, including systems-based, performance-based and risk-based approaches, that are:
- founded on science and based on risk, and that use common inspection procedures and tools;
- aligned with international standards, such as those developed by the Codex Alimentarius Commission, the World Organization for Animal Health (OIE) etc.;
- based on the premise that industry is responsible for its products and processes and must demonstrate ongoing compliance with legislative requirements; and
- flexible, to accommodate the complexity and size of an operation;
Regarding roles and responsibilities, regulated parties are responsible for complying with the law; they demonstrate this commitment by ensuring that the commodities and processes for which they are responsible meet regulatory requirements. In addition, they must provide the CFIA with input and information that is used in regulatory decision making. The CFIA verifies industry compliance through activities that include inspection, surveillance, sampling and testing. These activities are used to assess whether a regulated party has developed, documented, implemented and maintained written preventive control measures, whether these measures are effective, and whether regulatory requirements and permission conditions have been met. The CFIA also takes appropriate compliance and enforcement action when non-compliance is identified.
Risks posed by hazards in feeds must be managed or eliminated during production, processing, importing and distribution. It is the responsibility of regulated parties to:
- produce or import commodities that meet Canadian regulatory requirements;
- mitigate identified risks associated with those commodities; and,
- demonstrate a commitment to reducing risks associated with regulated feeds to an acceptable level through responsible practices such as:
- surveillance and control measures,
- processing controls (e.g., heat treatment, seed cleaning, bark removal, composting),
- cross-contamination controls (e.g., sanitation and hygiene, establishment of buffer zones or controlled access areas), or by
- limiting exposure (e.g., containment prior to processing).
The CFIA will take a common systematic approach to determining appropriate levels for oversight activities by assessing and re-assessing regulated parties' preventive control plans or relevant equivalents where required and the track record of regulated parties (including associated surveillance and recall data, certification to a relevant standard that is recognized by the CFIA, and the regulated party's compliance history).
The CFIA will regularly review, verify and/or update information to determine the overall level of risk presented. Based on the results of this process, the CFIA will determine the frequency, intensity and type of oversight activities required, and allocate its resources to those products and regulated parties that present the highest risk to Canadians, Canadian animal and plant populations and the environment. In allocating resources, the CFIA will also continue to take into account specific requirements of other countries to facilitate access to export markets.
A number of other triggers could also impact oversight activities by the CFIA and which can be included as elements of a risk management strategy. These include:
- responses to complaints, investigations, recalls or outbreaks/incursions in Canada;
- results of concern arising from an oversight activity, such as inspection or sampling
- information about a potential issue received from a third party (including, another government department, or an international trading partner);
- requests for inspection by a regulated party (e.g., for export marketing purposes); or,
- requests by a foreign trading partner or foreign delegation for a site visit.
The frequency, intensity or type of CFIA oversight activities may also be adjusted when:
- a regulated party implements and maintains a preventive control plan that exceeds minimum requirements; or
- the effectiveness of a regulated party's preventive control plan, in preventing and mitigating risks and achieving regulatory compliance, is consistently demonstrated through the regulated party's compliance history.
The Figure (below) illustrates the risk oversight process, highlighting the information and intelligence sources as well as the analyses used by the CFIA to determine the frequency, intensity and types of oversight activities appropriate in a given circumstance.
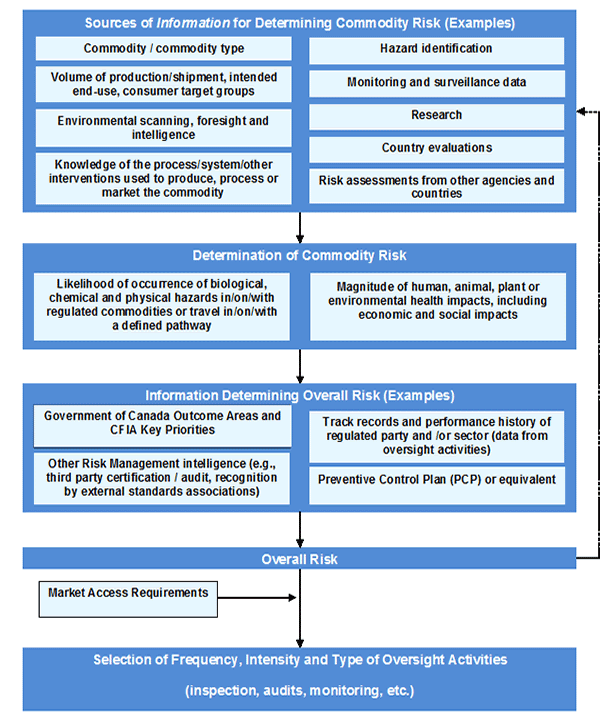
Description for Figure 1
Figure 1 illustrates the risk-based approach to the oversight process, highlighting the information and intelligence sources as well as the analyses used by the CFIA to determine the frequency, extent or type of regulatory oversight appropriate in a given circumstance.
A series of six connected horizontal rectangular boxes flow from top to bottom starting with
- Information for determining risk (examples), with an arrow pointing to
- Determination of initial risk, with an arrow pointing to
- Information for determining residual risk (examples), with an arrow pointing to
- Determination of residual risk, with an arrow pointing to
- Other information considered in risk management (examples), with an arrow to circle back to Information for determining initial risk as well as an arrow pointing to
- Selection of frequency, extent or type of oversight activities (e.g., inspection, monitoring, surveillance)
Rectangular box content
The first box, Information for determining initial risk, includes the following examples:
- Commodity/type/origin
- Volume of production/shipment, intended end-use, consumer target groups, destination
- Environmental scanning, foresight and intelligence
- Knowledge of the process/system/other interventions used to produce, process or market the commodity
- Hazard identification
- Monitoring and surveillance data
- Research
- Country evaluations
- Risk assessments by the CFIA, other agencies and countries
The second box, Determination of initial risk, reads as follows:
- Likelihood of occurrence of biological, chemical and physical hazards in/on/with regulated commodities or travel in/on/with a defined pathway
- Magnitude of human, animal, plant or environmental health impacts, including economic and social impacts
The third box, Information for determining residual risk, includes the following examples:
- Preventive Control Plan (PCP) or equivalent
- Track records and performance history of regulated party and /or sector (data from oversight activities)
There is no additional text associated with the fourth box, Determination of residual risk
The fifth box, Other information considered in risk management, includes the following examples:
- Government of Canada outcome areas and CFIA key priorities
- Other risk intelligence (e.g., private certification schemes / audit, recognition by external standards associations)
- Market access requirements
There is no additional text associated with the sixth box, Selection of frequency, extent or type of oversight activities
CFIA regulatory response to non-compliance
The CFIA has a mandate to respond to events that are initiated by lack of compliance on the part of the regulated parties and by events that are the responsibility of the Agency to control, such as pest and disease incursions. A compliance and enforcement strategy would be based on the principle that the regulated party is responsible for producing safe feed and healthy animals that comply with regulatory requirements. The corrective measures must be carried out by the regulated party. A response would be directed at the regulated party, including a licence holder, who does not comply with regulatory requirements. The response could include several activities as described in the CFIA's Compliance and Enforcement Operational Policy, such as product seizure and detention, suspension/cancellation of permissions, administrative monetary penalties and prosecution.
Support for Small Businesses
The CFIA is sensitive to the needs of small businesses and aware of the importance of finding the right balance between maximizing feed safety and minimizing administrative burden.
The following three elements of the proposed regulatory framework are deemed essential elements of the feed safety framework, regardless of organizational size:
- Permissions: So the CFIA knows who is doing what with feed for the purpose of oversight and, if necessary, enforcement. In addition, the CFIA would be able to reach out to small and medium businesses to ensure they are aware of their regulatory obligations, and to share compliance promotion information.
- Feed safety requirements: The proposed regulations would establish basic requirements for feed safety (e.g. sanitation, hygiene, pest control) with which everyone who manufactures, sells or imports feed would be required to comply.
- Traceability: The ability to trace a feed's path one step forward and one step back – to reduce the time it takes to locate and remove unsafe products from the market.
A "Small Business" is defined as …
… any business with fewer than 100 employees or between $30,000 and $5 million in annual gross revenues.
Given that the scope of the "small business" definition could still result in significant administrative burden on very small business with respect to the three (3) essential food safety framework elements identified above, a "micro-business" is further defined as follows for the purpose of this proposal:
"Any business with fewer than 5 employees and less than $30,000 in annual gross revenues" .
It is proposed that micro-businesses, at the time of application for a permission, be exempt from preparing a written PCP. The exemption would reflect the fact that these micro-businesses are generally less complex. However, the proposal is that this exemption will not be available for permission holders manufacturing feeds that are internationally recognized as having feed safety risks which require appropriate controls (i.e. medicated feeds, handling prohibited material). As risk ranking tools evolve, additional feeds could be excluded from the exemption through regulatory amendment.
In addition, the vast majority of small and micro-businesses would be given additional time to comply with certain requirements of the proposed regulations.
The CFIA will also develop a comprehensive compliance promotion strategy for small and medium businesses. Using the licensing information CFIA will be able to identify and reach out specifically to these businesses with plain language guidance material on how to comply with their regulatory obligations.
Finally, the CFIA will work with industry leaders and academic institutions to encourage a culture of feed safety, sharing of best practices in Canada and new opportunities for training.
Expected Coming into Force of the Proposed Regulations
The proposed regulations are expected to be published in Canada Gazette Part II by mid-2017.
The proposed new regulations represent a significant change to the way feeds are regulated in Canada. The CFIA recognizes that industry is at widely different levels of readiness:
- Those regulated parties that already belong to a feed safety program such as ANAC's FeedAssure, FAMI-QS, GMP+, or their own internal program are likely well positioned to meet the new regulatory requirements. Generally, these regulated parties are the larger businesses.
- Small and micro businesses are less likely to already have a preventive control plan in place and will likely have a higher compliance burden relative to the size of their business and may have a steeper learning curve.
- Importers, especially those who act as brokers, will have the greatest change in the requirements and are least likely to be ready to implement the full extent of the new regulations. Importers will now need to take on an additional level of responsibility for ensuring that they are sourcing safe and compliant products.
As such the Agency is proposing a staged implementation approach for the proposed regulations to come into force.
| Requirement | Large Businesses | Small Businesses | Importers | Micro Businesses |
|---|---|---|---|---|
| Permission(if applicable) | Upon coming into force of the regulations | Upon coming into force of the regulations | Upon coming into force of the regulations | Upon coming into force of the regulations |
| Written PCP | Upon coming into force of the regulations | One (1) year following coming into force of the regulations | One (1) year following coming into force of the regulations | Exempted |
Guidance Documents and Model Systems
The CFIA recognizes that good guidance documents will assist in the successful implementation of the proposed regulations. Guidance documents should facilitate compliance with regulations. The most important benefit of effective guidance and compliance promotion is improved feed safety through the effective implementation of preventive control measures.
Currently the CFIA has a range of guidance with respect to the existing Feeds Act and Regulations. While filled with important and useful information for industry, the existing guidance documents can be difficult to navigate to find the necessary information.
The CFIA is developing a new suite of regulatory guidance documents that will significantly improve the format, content and accessibility of its guidance material. This new suite of documents will replace existing manuals to help industry to comply with the Feeds Act and Regulations including:
- Guidance to clearly explain the intent of regulatory requirements.
- Model systems to provide examples that when properly applied have been demonstrated to achieve compliance with the regulatory requirements. These would initially include generic models.
- Additional "plain language" resources for small and medium size businesses.
The CFIA is intending to consult with regulated parties to determine the most appropriate format, language and methods of communicating guidance material.
The CFIA has also drafted a comprehensive Compliance Promotion Strategy which will apply to all regulated commodities (food, plant, feed, seed, etc.), the details of which are contained in a discussion document which is available on the CFIA website.
Model systems provide examples that when properly applied have been demonstrated to achieve compliance with regulatory requirements. These would initially be drawn from existing guidance and systems recognized by the CFIA, but in time could include other validated generic models or programs.
Role of 3rd Party Certification Programs
A number of external feed preventive control programs already exist. This includes the feed component of Canadian On Farm Food Safety Programs, ANAC's FeedAssure program, private industry programs, and a number of international programs such as GMP+, FAMI-QS, GLOBALG.A.P., and AFIA's Safe Feed/Safe Food program. The commitment to improving feed safety, shown by the number of parties that are choosing to participate voluntarily in one of these programs, is commendable and demonstrates the progressive role many in the feed industry are already playing. In addition to these existing programs, third parties – namely, industry-related non-government organizations, academia, or other service providers – have expertise and knowledge in their fields and may offer guidance, programs, training, self-assessments, and peer support to equip their members or sectors with the necessary tools to achieve compliance with the proposed changes to the Feeds Regulations. There are significant opportunities to leverage their leadership capacity and expertise through partnerships (e.g. private sector or public-private). Likewise, other government departments at the federal and provincial, and territorial levels may play an advisory role by providing expertise and tools.
Sector-specific domestic and international programs that include compliance with feed regulatory requirements as a specification may be considered as Model Systems and be taken into consideration by CFIA in the design and delivery of risk-based inspection oversight programs.
User Fees Modernization
When the CFIA was created in 1997, the intent of Parliament was that services would be provided on a cost recovery basis. Upon creation, the CFIA retained the cost recovery approaches of its parent organizations, including user fees for the feed program. These fees have not been updated since the Agency was created due to a moratorium on new and increased fees, that was in place until 2009. The moratorium has impeded the CFIA's ability to align fees with escalating program delivery costs, shifts in demand, changes in technology, new programming and evolving government policies relating to user fees and service standards. Since the moratorium was lifted in 2009, the CFIA has been reviewing its service standards and user fees to better align fees with the cost to deliver services where private benefits are derived.
Concurrently with the Agency's feed regulatory modernization initiative, the CFIA is reviewing its services, user fees and service standards for the feed program, in order to bring them into line with the new design and regulations. This puts the focus of cost recovery on services that the CFIA is delivering and the resources it is expending, rather than the the products that are involved or the size of the operation of the service recipient. The CFIA's intent is to move towards a fee structure that is reflective of the cost to provide its services and allows for service recipients to be charged appropriately for the services they use. The fee setting process encompasses four main steps:
- costing;
- assessment of public and private benefits;
- calculating the upper limit of the fee level (fee ceiling); and
- consideration of other relevant factors, including international benchmarking of relevant jurisdictions that provide comparable services to the CFIA.
The service standards for individual services will be based on current service delivery targets, and where relevant, to service standards Canada is committed to under international agreements. Information on user fees and service standards, once finalized, will be reported annually in the Agency's Departmental Performance Report.
Separate consultations on the proposed user fees for these services will occur once the review is completed. Stakeholders will have the opportunity to provide feedback on the proposed user fees and service standards at that time.
Inspector Training
The need for a comprehensive training system to support Agency transformation and modernization of the CFIA was the focus of many stakeholder comments during the preliminary consultations.
The CFIA is building a Learning and Training Architecture Forward Plan 2013-2019 (Plan) to support a modernized Agency. Currently, CFIA training reflects a commodity-specific inspection approach, and paper-based record keeping and interactions with stakeholders. The proposed Plan moves the CFIA to a single inspection approach consistent across commodities, supported by standardized training, technology information solutions, enhanced proactive science capacity and improved services to stakeholders.
To support this initiative, CFIA will undertake three key activities in responding to Agency Transformation, including:
- Design and development of new science and technical training initiatives, including training on the Feeds Act and the proposed regulations;
- Delivery of refresher training to current CFIA inspectors; and
- Development of the CFIA Learning and Training Architecture. This may include activities such as trainer qualification, e-learning design and development, virtual learning, and classroom learning.
CFIA has also been looking at training best practices around the world – one that stands out is the International Food Protection Training Institute (IFPTI) based in Battle Creek, Michigan. The IFPTI is the training initiative of the Global Food Protection Institute (GFPI). GFPI is a model for private-public partnerships that addresses issues related to food protection. The IFPTI collaborates with industry, academia, federal and state government, and other national and state organizations to develop effective and relevant materials that advance food protection.
The IFPTI has produced a National Curriculum of evidence-based, career-spanning training for food protection officials. This National Curriculum supports a fully integrated national food safety system as outlined in the U.S. Food and Drug Administration's Food Safety Modernization Act. Under the new private-public partnership, Safe Food Canada – The Learning Partnership, the CFIA is collaborating with key stakeholders from industry, academia and provincial/territorial governments to coordinate a Canadian effort to explore the potential of creating a model system similar to IFPTI in Canada to operationalize national training and certification in support of a professional and competent workforce. In following the IFPTI model, the development of a consistent curriculum framework for regulatory compliance and enforcement has the potential to standardize the professional development of federal, provincial and local feed regulators in Canada and all employees working in the feed industry.
Conclusion
This proposal presents in greater detail the policy direction intended by the proposed regulations, to give stakeholders another opportunity to provide feedback before the proposed regulations enter into the formal consultation process.
The proposed regulations will represent a significant stride in augmenting regulated industry's responsibility and a clear raising of the bar for feed safety in Canada via the application of hazard identification and preventive.
In circling back to the feed regulatory modernization objectives that were established for this project:
Develop a modernized risk-based and outcome-based regulatory framework for feeds which:
- safeguards feeds and the food production continuum;
- attains the most effective and efficient balance between fair and competitive trade in the market; and
- minimizes regulatory burden,
the following summary identifies how the proposal serves to accomplish them.
| Key Result | How the modernized framework proposes to achieve objective … |
|---|---|
| Modernized Feeds Act |
|
| Modernized, Integrated Agency Inspection Model (iAIM) |
|
| Significant reduction in pre-market product registration |
|
| Key Result | How the modernized framework proposes to achieve objective … |
|---|---|
| Focus of regulations on health and safety, prevention or mitigation of risks to public, animal and plant health, the environment |
|
| Enable regulated parties and CFIA to rapidly respond and mitigate emerging feed and food safety risks |
|
| Key Result | How the modernized framework proposes to achieve objective … |
|---|---|
| Reflect changes in trade, science and technology |
|
| Timely updating of feed safety and other standards |
|
| Aligning, to the extent possible, with international trading partner and international standards |
|
| Key Result | How the modernized framework proposes to achieve objective … |
|---|---|
| Significant reduction in pre-market product registration |
|
| Feed labelling |
|
The coming years will be a time of change, challenge, and opportunity as Canada moves forward in the development and implementation of the proposed regulations that will strengthen protection of Canada's feed and food supply.
These proposed regulations will not be the final step in the evolution of Canada's regulatory framework under the Feeds Act. Rather, an effective feed safety control system is marked by ongoing policy review and continuous improvement. The CFIA is committed to such a process and welcomes feedback on future improvements, as well as those addressed in this round of regulatory change.
Next Steps
The CFIA welcomes feedback from stakeholders and the public on the proposed regulatory changes presented in this document.
Written comments regarding any element of the proposed regulations should be forwarded by April 8, 2016 to:
Animal Feed Division
Canadian Food Inspection Agency
59 Camelot Dr.
Ottawa, Ontario
K1A 0Y9
Comments can also be emailed to Serge Tolusso
Comments received by April 8, 2016 will be taken into consideration prior to publication of the proposed regulations in Canada Gazette Part I.
Annex 1: What We Heard from Consultations
The CFIA has prepared and distributed several subject-specific proposals to date for stakeholder review and comment regarding the modernization of the feed regulatory framework. In response to stakeholder feedback to each proposal, the CFIA has also prepared individual reports to summarize the feedback received and provide responses to it. A listing of and links to the summary reports prepared to date follows below.
Feed Ingredients
Feed Labelling
Collective Ingredient Terms Proposal – Consultation Summary – Respondent Comments and CFIA Responses
Feed Hazard Identification/Preventive Controls
The collective feedback to all these individual consultations has been taken into careful consideration in the preparation of this consolidated framework proposal.
Annex 2: Proposed Labelling Requirements
| Component | Current Requirement | Proposed |
|---|---|---|
| Manufacturer Name & Address | In the case of a feed not required to be registered, the name and address of the person who manufactured the feed or caused it to be manufactured | Name and address of manufacturer or who caused it to be manufactured (which includes allowing a head office contact) for feeds that are not registered. Additional names and addresses such as those of the head office for the company, importer or distributor may also be included on the label on a voluntary basis. |
| Registrant Name & Address, Reg'n # | In the case of a feed required to be registered, the name and address of the registrant | For feeds that are registered, name and address of the registrant. May add the manufacturer information on a voluntary basis. |
| Feed Name | The name of the feed in accordance with section 32 The name of a feed shall be appropriate for the intended use of the feed and shall not be misleading. |
The feed name should reflect the following components: Purpose or Feed Type Species or Category of Intended Livestock |
| Brand Name | The brand of the feed, if any | The brand of the feed, if any |
| Net Amount | The net amount expressed as the number of units in a package, in the case of a package of feed containing individual feeding forms, or Expressed as the mass or volume in the package or shipment, in the case of any other package or bulk shipment of feed |
The net amount expressed as the number of units in a package, in the case of a package of feed containing individual feeding forms, or Expressed as the mass or volume in the package or shipment, in the case of any other package or bulk shipment of feed |
| Registration # | The registration number, where applicable | Must have registration number, can be anywhere on label provided it's "conspicuous, legible, indelible, etc" |
| Guaranteed Analysis | An accurate statement of the guaranteed analysis in respect of the feed (see Table 3 for required guarantees) The label of a feed shall not show a guarantee for a nutrient that is not required to be guaranteed unless.. |
It is proposed that the same approach continue to be taken and we will further examine the requirements for specific feed types. Will examine Table 3 and make any needed changes. |
| Directions for Use | Subject to section 27(3) [any directions for use required to be shown on a label may refer to an insert…if insert is enclosed within the package…] directions for use in sufficient detail to permit safe and effective use of the feed for its intended purpose by users with no special knowledge of the purpose and use of the feed. | Require directions for use for all feeds. Guidance (outside of regulations) to be provided to allow a purpose statement or a "for use in livestock feeds" as sufficient directions for use. |
| List of Ingredients | In the case of a feed that is required to comply with the guarantees set out in item 7 of Table 3 of Schedule I, the name of each ingredient in the feed In the case of a feed other than a feed referred to [above] the name of each ingredient in the feed or the statement "a list of ingredients used in this feed may be obtained from the manufacturer of registrant." |
All feeds require the complete list of ingredients for that feed on the label Collective terms allowed. Manufacturer/registrant must supply actual list of ingredients by their Sched IV names upon request. |
| Lot # | An identification code, in the case of a micro-premix feed or a feed designed to replace whole milk in the ration of the livestock. | All feeds must have a lot number. For further guidance/discussion: Must be clearly identifiable as a lot #. Manufacturer must be able to confirm when and where the product was manufactured. |
Medicated Feeds:
|
If the feed is a medicated feed, other than a veterinary prescription: The name and actual amount of the medicating ingredient present in the feed, in accordance with the CMIB, in direct association with the feed name The claim or claims applicable with the kind of medicating ingredient present in the feed, the level of MI in the feed and the type of livestock for which the feed is intended, as set out in the CMIB Every caution statement in respect of the MI present in the feed that is set out in the CMIB under or next to the heading, in bold print "caution" . Each statement shall be clearly separated from any other caution statement and any other information on the label. Every warning statement in respect of the MI present in the feed that is set out in the CMIB under or next to the heading, in bold print "warning" . Every warning statement shall be clearly separated from any other warning statement and any other information shown on the label. |
If the feed is a medicated feed, other than a veterinary prescription: The name and actual amount of the medicating ingredient present in the feed, in accordance with the CMIB, in direct association with the feed name The claim or claims applicable with the kind of medicating ingredient present in the feed, the level of MI in the feed and the type of livestock for which the feed is intended, as set out in the CMIB Every caution statement in respect of the MI present in the feed that is set out in the CMIB under or next to the heading, in bold print "caution" . Each statement shall be clearly separated from any other caution statement and any other information on the label. Every warning statement in respect of the MI present in the feed that is set out in the CMIB under or next to the heading, in bold print "warning" . Every warning statement shall be clearly separated from any other warning statement and any other information shown on the label. |
| Form | If the feed contains a medicating ingredient and is in a form other than a mash, the particular form of the feed | If the feed contains a medicating ingredient and is in a form other than a mash, the particular form of the feed |
| Claims | A label shall not contain…any claim for a mixed feed or any ingredient in the feed unless evidence has been assessed or evaluated and found to support the claim. …any claim for a single ingredient feed unless evidence has been assessed or evaluated and found to support the claim. |
It is proposed that the basic approach for claims should be that labels should not contain claims other than those which are truthful and verifiable. Also proposed that not all products with label claims will require premarket assessment. Proposed development of a list of permissible claims which would not trigger a requirement for registration. Claim would still need to be assessed and approved can be done via policy not registration. |
| Units of Measurement | All units of measurement shown on a label of a feed shall be expressed in metric measure only in accordance with the Weights and Measures Act. | Only metric, except allow net weight to appear in imperial. For multi-jurisdictional label create a specific exemption which would allow other units to meet the requirements of those other markets. |
| PM statement | If the feed contains pm as defined in HoA, the following statement written legibly, indelibly and conspicuously: "Feeding this product to cattle…" |
If the feed contains pm as defined in HoA, the following statement written legibly, indelibly and conspicuously: "Feeding this product to cattle…" |
| Consultant formula | In the case of a consultant formula feed, the name and address of the specific purchaser for whom the feed was manufactured. | Based on changes to what requires registration this class of feed should not be required |
| SIF | In addition to the labelling requirements prescribed in subsection (1), a single ingredient feed shall be labelled in accordance with the labelling requirements set out for that feed in Schedule IV. | In addition to the labelling requirements prescribed in subsection (1), a single ingredient feed shall be labelled in accordance with the labelling requirements set out for that feed in Schedule IV. |
| Container (place of manufacture) | Where any reference, direct or indirect, is made on a label or container to the place of manufacture of the label or container and not to the place of manufacture of the feed, the reference shall be accompanied by an additional statement indicating that the place of manufacture refers only to the label or container. 26(9) | Where any reference, direct or indirect, is made on a label or container to the place of manufacture of the label or container and not to the place of manufacture of the feed, the reference shall be accompanied by an additional statement indicating that the place of manufacture refers only to the label or container. 26(9) |
| Place of manufacture | Where a feed that is manufactured or produced and labelled in a country other than Canada has applied to it a label that shows the identity and principal place of business of the person in Canada for whom the feed was manufactured or produced for resale, the identity and principal place of business of that person shall be preceded by the words "imported by" or "imported for", unless the geographic origin of the product is stated on the label. | Where a feed that is manufactured or produced and labelled in a country other than Canada has applied to it a label that shows the identity and principal place of business of the person in Canada for whom the feed was manufactured or produced for resale, the identity and principal place of business of that person shall be preceded by the words "imported by" or "imported for", unless the geographic origin of the product is stated on the label. |
| Biological testing claim | No statement shall be made on the label of a feed indicating that such feed has been biologically tested unless The feed has been biologically tested using scientifically acceptable test methodology The results of such tests supports any claim contained in the statement or any claim implied by the statement Evidence that such tests have been made and the results of such tests have been supplied to the Director for assessment or evaluation. |
No statement shall be made on the label of a feed indicating that such feed has been biologically tested unless The feed has been biologically tested using scientifically acceptable test methodology The results of such tests supports any claim contained in the statement or any claim implied by the statement Evidence that such tests have been made and the results of such tests have been supplied to the Director for assessment or evaluation. Should work with the Permissible Claims concept (e.g. Director's approval is via adding claim to the list). |
| Language | Any information required to be shown on the label of a feed shall be printed conspicuously, legibly and indelibly in English or French or both languages. | Proposed that any label information which impacts health and safety will be required to be present in both French and English (to meet Official Languages requirements). See "international labels" document for full details on allowing other languages on labels. Allow the geographical exemption but require that the manufacturer provide the label information in the language of choice. |
| Location of information | Subject to subsection (3) and unless otherwise authorized by the Director, any information that is required to be shown on the label shall appear on the main panel of the package label or where the package contains less than 5 kg on the surface of the package. (3) Any directions for use required to be shown on a label may refer to an insert containing detailed directions for use if such an insert in enclosed within the package that bears the label. |
Use "principal display surface" definition from CPLR. Feed regs will specify which info is required in principal display surface and which may be listed elsewhere |
| Caution and warning statements | Currently as part of medicated feed requirements, feed ban, or ingredient labelling. | Same as current plus… Labels would also require any additional cautions or warnings which convey useful information to the purchaser. Proposed that when a feed is manufactured using an ingredient or mixed feed which has a caution or warning statement on its label, or for which there is a maximum inclusion rate of an ingredient, this relevant information must be transferred onto the label for the new feed product. |
| Expiry/Best Before dates | No current requirement. Allowed or required on some registered feeds and verified via registration | Proposed that for feeds which do not require a "best before" or "expiry" date, one may be indicated on the label on a voluntary basis. |
Annex 3: Written PCP
The proposed requirements for a written PCP could be reflected in the regulations as follows:
A Preventive Control Plan (PCP) means in respect of a food or the manufacturing, preparing, storing, packaging, labelling, growing, harvesting and importing of a food, a written preventive control plan that effectively:
- Identifies for Elements 1-7:
- all hazards that must be prevented, eliminated or reduced;
- critical control points and related control measures that are validated by evidence that demonstrates that the control points and related control measures effectively control the hazards identified;
- critical limits;
- monitoring procedures;
- corrective action procedures;
- verification procedures; and
- record keeping procedures; and
- Identifies the measures they have taken to comply with other relevant regulatory requirements, including those FDR requirements that will be incorporated in the proposed regulations e.g. packaging and labelling provisions of FDR.
Every licence holder and every person who grows or harvests a fresh fruit or vegetable that is to be sent or conveyed from one province to another must:
- prepare, keep and maintain a PCP;
- implement the PCP;
- prepare, keep and maintain documents that substantiate that the food has been imported, manufactured, prepared, stored, packaged, labelled, grown and harvested in accordance with the PCP;
- prepare, keep and maintain documents that substantiate that the manner in which the food was imported, manufactured, prepared, stored, packaged, labelled, grown and harvested is in compliance with the Act and the Regulations; and
- prepare, keep and maintain a document that sets out a procedure for the maintenance and review of the PCP, and implement that procedure.
Every person who prepares a food that is to be exported and every person who grows or harvests fresh fruits and vegetables that is to be exported must prepare, keep, maintain and implement a PCP as described above if they request an export certificate.
- Date modified: