D-11-04: Canadian Compliance Program for Grain Screening Pellets (GSP) to meet the phytosanitary import requirements of the United States
This page is part of the Guidance Document Repository (GDR).
Looking for related documents?
Search for related documents in the Guidance Document Repository
Effective Date: February 17, 2012
(Original)
Subject
Grain screening pellets (GSP) exported to the United States (U.S.) for all end uses other than as fuel, must be produced in a Canadian Food Inspection Agency (CFIA) approved facility. This directive describes: 1) the process for Canadian processing facilities to apply and be approved by the CFIA to manufacture GSP for export to the U.S., and 2) the specific requirements that must be met in the production of GSP in Canada to meet the United States Department of Agriculture's (USDA) phytosanitary import requirements.
Table of Contents
- Review
- Endorsement
- Amendment Record
- Distribution
- Introduction
- Scope
- References
- Definitions, Abbreviations and Acronyms
- 1.0 General Requirements
- 2.0 CFIA GSP Program Requirements
- 3.0 Responsibilities
- 4.0 Non-Compliance
- 5.0 Appendices
- Appendix 1 - Compliance Agreement Between Canadian Facilities Manufacturing Grain Screening Pellets for Export to the United States. and the Canadian Food Inspection Agency
- Appendix 2 - Inspection Checklist - Canadian Compliance Program for Grain Screening Pellets to the United States
- Appendix 3 - Example of Approval Letter
- Appendix 4 - Example of Export Certificate Issued by the Approved Pelleting Facility
Review
This directive will be reviewed every 5 years unless otherwise needed. For further information or clarification, please contact the CFIA.
Endorsement
Approved by:
![]()
Chief Plant Health Officer
Amendment Record
Amendments of this directive will be dated and distributed as outlined in the distribution below.
Distribution
- Directive mail list (CFIA, USDA, etc.)
- Provincial Governments, Industry (determined by author)
- National Industry Organizations (determined by author)
- Internet
Introduction
In November of 2009 the USDA, Plant Protection and Quarantine (PPQ) established phytosanitary import requirements that prohibited the importation of GSP from Canada intended for use as livestock feed. The restrictions were implemented over concerns that GSP had the potential to act as a pathway for the movement into the U.S. of viable regulated weed seeds included on the USDA's Federal Noxious Weed Seed List. To address these concerns, the CFIA has developed a compliance program for Canadian GSP processing facilities, which is recognized by the USDA. The requirements for this compliance program are based on the process used by the USDA to approve facilities located in the U.S. that currently import unprocessed grain screenings of non approved crop kinds from Canada.
To export GSP from Canada into the U.S., a compliance agreement between the processing facility and the CFIA must be in place. Under this agreement GSP being exported from Canada to the U.S. must
- be manufactured from Canadian and/or U.S. origin screenings
- meet the requirements of Section 2.0 of this directive.
- be accompanied by an export certificate issued by the approved facility that attests to the origin, treatment and crop kinds used to produce the GSP.
An agreement of this type will meet the import requirements of the USDA.
Scope
The information in this directive is intended for CFIA inspectors and program officers and grain elevators or facilities that manufacture or export GSP to the U.S.
References
United States Department of Agriculture/Animal and Plant Health Inspection Services (USDA/APHIS) Canadian Border Agricultural Clearance Manual 04/2006-37
Definitions, Abbreviations and Acronyms
Definitions for terms used in the present document can be found in the Plant Health Glossary of Terms.
1.0 General Requirements
1.1 Legislative Authority
Plant Protection Act, S.C. 1990, c. 22
Plant Protection Regulations. SOR 95/212
Canadian Food Inspection Agency Fees Notice, Canada Gazette, Part 1 (as amended from time to time)
1.2 Fees
The CFIA is charging fees in accordance with the Canadian Food Inspection Agency Fees Notice. Anyone requiring other information regarding fees may contact any local CFIA office or visit us at our Fees Notice Web Site.
1.3 Regulated Pests
Viable seeds of all weeds listed under 7 CFR 361.6, including both Federal Noxious Weeds and weeds with tolerances applicable to their introduction. This list can be found at the following link. https://plants.usda.gov/java/noxious?rptType=Federal
1.4 Regulated Commodities
GSP produced from Canadian and/or U.S. origin grain screenings derived from approved crop kinds in a Canadian facility for export to the U.S. for use in or as livestock feed.
Table 1 - Approved crop kinds are:
- barley
- chickpeas
- flax
- proso
- broomcorn
- cowpeas
- lentils
- rye
- buckwheat
- field corn
- millet
- sorghum
- canary seed
- field beans
- mustard
- soybeans
- canola
- field peas
- oats
- wheat
1.5 Exempt Commodities
Pellets made from the residual matter remaining after the oil has been extracted from the grain.
Pellets consisting of a mixture of various grains that have been ground to a uniform powder-like consistency.
Pellets exported for use as fuel and accompanied by an end use statement to that effect.
1.6 Prohibited Commodities
GSP produced from unapproved crop kinds not listed in Table 1 above.
GSP imported into Canada from any country other than the U.S.
GSP produced from material that originates from any country other than Canada and/or the U.S.
1.7 Regulated Areas
All areas of Canada
2.0 CFIA GSP Program Requirements
2.1 Application Requirements
Processors who manufacture GSP for export to the U.S. must apply to participate in this compliance program by completing the compliance agreement (Appendix 1) and forwarding it to their local CFIA office.
2.2 CFIA Inspection Requirements
The CFIA will conduct:
- an initial inspection of the facility prior to approval to evaluate the systems in place used in the manufacturing of GSP for export to the U.S.
- ongoing annual inspections to verify compliance with this program.
The CFIA retains the option to visit the facility at any time to verify compliance or to take official samples of the GSP and submit these to a CFIA or CFIA approved lab for testing. If requested, the CFIA may conduct joint inspection(s) of any approved facilities with USDA inspection staff.
2.3 GSP Facility Inspection Requirements
GSP facilities must:
- Cooperate with CFIA during inspections and allow access to records as required to verify compliance with this program.
- Maintain records for 2 years listing the crop type(s) and their origin(s) used in the production of GSP for export to the U.S.
- Demonstrate that the grain screenings used in the manufacture of GSP for export are produced from eligible crops that have been grown in Canada or the U.S.
- Ensure bins and conveyances (i.e. trucks or railcars) were emptied of other products prior to loading GSP manufactured under the compliance program to avoid contamination with other products.
- demonstrate that grain screenings used in the manufacturing of the GSP are put through a hammer mill with a maximum screen size of 2.38 millimetres (6/64 of an inch).
- demonstrate that during the manufacturing of GSP the heat used in the pelleting process was maintained at a minimum temperature of 85 degrees Celsius (185 degrees Fahrenheit).
2.4 GSP Product Testing Requirements
Samples of GSP will be collected by a CFIA inspector during the initial inspection and subsequent annual inspections and submitted for analysis to a CFIA or CFIA approved lab and tested according to USDA GSP lab testing protocols. Samples will be tested to verify all seeds, including the U.S. Federal Noxious Weed Seeds listed under 7 CFR 361.6 have been devitalized.
2.5 Results of Inspection
Once a facility has been inspected and samples have been tested according to the requirements of Section 2.4, the facility will receive a copy of the approval letter from the CFIA (Appendix 3). Within this letter, the facility will be issued a unique facility approval number (CFIA-GSP-XX). The facilities name, address and the CFIA issued approval number will be shared with the USDA. Approved facilities will be identified as "approved for export" in the USDA/APHIS Canadian Border Agricultural Clearance manual in the section related to imports of grain screening pellet imports for end users other than fuel.
3.0 Responsibilities
3.1 CFIA Responsibilities
The CFIA regional program officer or inspector is responsible for:
- evaluating and approving the compliance agreement submitted by the facility;
- facility inspections;
- sampling and submission of GSP samples for lab testing to a CFIA or CFIA approved lab;
- obtaining a unique facility approval number from the CFIA-Grains and Oilseed Section in Ottawa and assigning it to the facility;
- writing the approval letter (Appendix 3).
A signed copy of the compliance agreement and the approval letter will be sent to the National Manager, Grains and Oilseeds Section in Ottawa and to the Area Program Specialist. The CFIA-Grains and Oilseed Section will inform the USDA as GSP processing facilities are approved or removed from this compliance program so that the USDA/APHIS Canadian Border Agricultural Clearance Manual is up to date.
3.2 Approved GSP Facility Responsibilities
The facility is responsible for ensuring that all GSP produced for export to the U.S. meets the requirements listed in Section 2.3.
The facility shall issue an Export Certificate (Appendix 4) for each shipment specifying the facility name and CFIA issued approval number, the crop type(s) and origin(s) of the grain screenings and bin number(s) the GSP were shipped from. A shipment identification number (i.e. Bill of Lading Number) must be included on each Export Certificate. The facility will maintain copies of the certificates issued along with records of the signed compliance agreement with the CFIA (Appendix 1) and the approval letter (Appendix 3).
4.0 Non-Compliance
The CFIA may suspend or revoke a facility's approval status upon finding non-compliance with the requirements specified in this directive.
5.0 Appendices
Appendix 1: Compliance Agreement between Canadian Facilities Manufacturing Grain Screenings Pellets for Export to the United States and the Canadian Food Inspection Agency
Facility Name: ![]()
Facility Contact: ![]()
Address: ![]()
Telephone No.: ![]()
Fax: ![]()
This agreement certifies that the Grain Screening Pellets (GSP) manufactured by the above facility for export to the United States (U.S.) were produced according to the requirements specified in D-11-04, Canadian Compliance Program for GSP to meet the phytosanitary import requirements of the U.S.
The above facility shall:
- Allow the Canadian Food Inspection Agency (CFIA) to conduct inspections, as required, to verify compliance with this agreement.
- Maintain records for two years including the crop type(s) and their origin(s) used in the production of GSP for export to the U.S.
- Demonstrate that the grain screenings used in the manufacture of GSP for export to the U.S. are only of screenings of eligible crop kinds grown in Canada or U.S.
- Maintain a current written protocol describing the GSP manufacturing process employed by the facility and ensure it meets the specific temperature and milling requirements as outlined in Section 2.3 of D-11-04.
- Ensure bins and conveyances (i.e. trucks or railcars) were emptied of other products prior to loading GSP manufactured under the compliance agreement to avoid contamination with other products.
- Include an export certificate with all shipments of GSP specifying the facility name and CFIA issued approval number. The certificate must also identify the crop type(s) and origin(s) of the grain screenings and the bin number(s) from which the GSP were shipped from. A shipment identification number (i.e. Bill of Lading Number) must be included on each certificate. In addition, the facility must maintain records of their compliance agreement with the CFIA and their CFIA approval letter.
Further, I am and shall be responsible for and shall indemnify and save harmless Her Majesty the Queen in Right of Canada, including Canadian Food Inspection Agency, Her officers, Employees, Successors and Assigns, from and against all manners of actions, causes of action, claims, demands, loss, costs, damages, actions or other proceedings by whomsoever made, sustained, brought or prosecuted in any manner based upon, caused by, arising out of, attributable to or with respect to any failure, inadvertent or otherwise, by act or omission, to fully comply with the said terms, conditions and requirements.
Dated ![]() , 20
, 20![]()
![]()
Signature - Facility Contact
![]()
Print Name
Facility Recommended for Approval by:
![]()
CFIA Inspector (Sign and Print)
![]()
Date
GSP Product Sample Testing Completed and Product Recommended for Approval:
Yes ![]() No
No ![]()
Approved for Participation in the Canadian Compliance Program for Grain Screening Pellets:
![]()
CFIA Regional Program Officer (Sign and Print)
Canadian Food Inspection Agency
![]()
Date
CFIA Assigned Facility Approval Number CFIA-GSP- ![]()
Appendix 2: Inspection Checklist - Canadian Compliance Program for Grain Screening Pellets to the U.S.
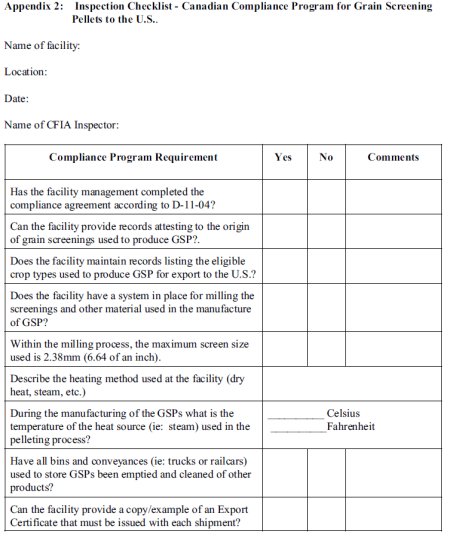
Description - Inspection Checklist - Canadian Compliance Program for Grain Screening Pellets to the U.S.
This is description of the inspection checklist - Canadian Compliance Program for Grain Screening Pellets to the U.S.
Some of the requirements for this inspection are:
- Has the facility management completed the compliance agreement according to D-11-04?
- Can the facility provide records attesting to the origin of grain screenings used to produce GSP?
- Does the facility maintain records listing the eligible crop types used to produce GSP for export to the U.S.?
- Does the facility have a system in place for milling the screenings and other material used in the manufacture of GSP?
- Within the milling process, the maximum screen size used is 2.38mm (6.64 of an inch).
Appendix 3: Example of Approval Letter
[print on CFIA letterhead]
[Company name and address]
[date]
Dear [name],
An inspection of (enter facility name and location here) on, (enter date) has confirmed that your facility meets the requirements as outlined in CFIA's directive D-11-04: Canadian Compliance Program for Grain Screening Pellets (GSP) to meet the phytosanitary import requirements of the United States.
By way of this letter, I am approving (enter facility name and location here) to produce grain screening pellets for export to the U.S.
As outlined in your compliance agreement signed (DATE) and submitted to your local CFIA office, your facility will be subject to an annual inspection should you wish to maintain your approval under this program.
The unique CFIA issued number that has been assigned to your facility is:
CFIA - GSP - ![]() .
.
If you have questions or concerns regarding this compliance program , please do not hesitate to contact me.
Yours truly,
Regional Program Officer, Plant Protection
CC National Manager, Grains and Oilseeds Section, CFIA
Area Grains and Oilseeds Program Specialist, CFIA
Appendix 4: Example of Export Certificate Issued by the Approved Pelleting Facility
Mandatory title for use on all certificates: "Export Certificate Under CFIA Directive D-11-04"
The following information fields and attestation below must appear on the certificate:
Shipment Identification Number ![]()
Facility Name
Facility Address
CFIA Facility Approval Number - CFIA - GSP -![]()
Bin Number(s) -
This certificate attests that the grain screening pellets in this consignment are not intended to be used for planting, and;
- meet the processing requirements as outlined in the compliance agreement between the above facility and the CFIA as per Directive D-11-04.
- have been produced from grain screenings of either U.S. or Canadian origin.
- have been produced from one or more of the following Canadian or U.S. grown crop kinds:
Name:
Signature:
Date:
- Date modified: