Bretziella fagacearum (previously known as Ceratocystis fagacearum (Oak Wilt) - Fact Sheet
Identity
Name: Bretziella fagacearum (Bretz) Z.W. de Beer, Marinc., T.A. Duong and M.J. Wingf (new scientific name used by the CFIA as of November 2018)
Synonym(s): Chalara quercina Henry, Ceratocystis fagacearum (Bretz) Hunt
Anamorph: Endoconidiophora fagacearum Bretz
Taxonomic Position: Fungi: Ascomycetes: Ophiostomatales
Common Name(s): Oak Wilt, Flétrissement du chêne
Main Disease
Oak wilt is caused by the fungus Bretziella fagacearum which develops in the outer sapwood of the tree. The infected tree reacts by developing tyloses and gums which subsequently restrict the flow of water and nutrients in affected vascular tissues with the result that the tree wilts and subsequently dies (French & Stienstra, 1980).
Host Range
All species and varieties of oak (Quercus spp.) tested have been found to be susceptible to oak wilt, although species in the red oak group are more seriously affected (French & Stienstra, 1980). Quercus species in the red oak group are usually killed within one year of becoming infected, whereas white oaks frequently recover from infection or take many years to die.
The following species are recorded as hosts (Farr et al., 1989; Liese & Ruetze, 1987):
- Quercus alba (white oak)
- Quercus coccinea (Scarlet oak)
- Quercus ellipsoidalis (northern pin oak)
- Quercus falcata (southern red oak)
- Quercus fusiformis (live oak)
- Quercus ilicifolia (bear oak, scrub oak)
- Quercus imbricaria (laurel oak)
- Quercus macrocarpa (bur oak)
- Quercus marilandica (blackjack oak)
- Quercus muehlenbergii (Chinkapin oak)
- Quercus nigra (possum oak, water oak)
- Quercus rubra (red oak)
- Quercus palustris (pin oak)
- Quercus phellos (willow oak)
- Quercus prinus (chestnut oak)
- Quercus rubra (northern red oak)
- Quercus rubra var. borealis (northern red oak)
- Quercus stellata (white post oak)
- Quercus texana (Spanish oak, Texas red oak)
- Quercus velutina (black oak)
- Quercus virgniana (southern live oak)
Several varieties of apple (Malus), both American and European chestnuts (Castanea), species of chinquapin (Castanopsis) and tanoak (Lithocarpus) have been demonstrated to be susceptible to B. fagacearum following artificial inoculation (Sinclair et al., 1987; Rexrode & Brown, 1983). Bretz and Long (1950) reported isolating the oak wilt fungus from naturally infected Chinese chestnuts (Castanea mollissima), and Rexrode & Brown, 1983 subsequently reported plantation-grown trees to be infected.
Distribution
- North America: USA (AL, IL, IN, IA, KS, KY, MD, MI, MN, MS, MO, NE, NC, OH, OK, PA, SC, SD, TN, TX, VA, WV, WI).
Biology
Oak wilt is a vascular disease, meaning the fungus is usually found only in the vascular tissues of the outermost xylem. In the case of diseased red oaks, fungal spores may be transported to all parts of the tree as the tree nears death, although rarely in small branches less than 3-4" in diameter (French & Stienstra, 1980). Bretziella fagacearum has been isolated from the xylem of roots, trunk, branches, twigs and even leaf petioles of diseased red oaks (Gibbs & French, 1980). In white oaks, the fungus' distribution is more restricted, occurring only in the xylem of the current year's growth. If the tree recovers, annual growth buries the infection under new xylem with the result that old infections do not serve as a significant source of inoculum (French & Stienstra, 1980; Gibbs & French, 1980).
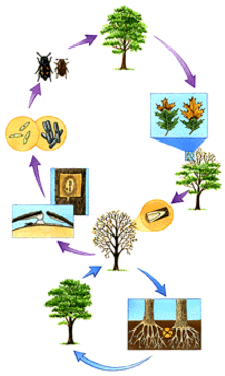
Above-ground: Nitidulid beetles or bark beetles carry spores of the fungus from spore mats on infected trees to wounds on healthy trees during feeding or breeding
Below-ground: The fungus travels through the interconnected roots of infected and healthy trees growing in close proximity.
When a diseased red oak dies, the fungus develops sporulating mats in the bark which produce conidia and ascospores when mature. These mats vary in size but range from approximately 1-8 inches × ½-4 inches to as large as 13 inches long and 7 inches across (French & Stienstra, 1980). Conidia are produced in large numbers, but ascospores are only produced by when two compatible strains of the fungus are present. These fungal mats are produced only on red oaks, and are not seen on live oaks or white oaks (Davies, 1992). They are generally produced in late fall or early spring and are viable for two to three weeks only, attracting insects which crawl over, burrow into and feed on them. Sticky spores adhere to the insects and are subsequently carried to healthy trees where they are deposited in wounds when the insects alight (Davies, 1992). Sporulating mats have been observed primarily on trunks and large branches, but there are reports of mats being produced on branches as small as 2 - 4 cm diameter (Gibbs & French, 1980). In the southern parts of the range of oak wilt, sporulating mats occur only rarely and dissemination occurs more often as a result of the activities of oak bark beetles.
Bretziella fagacearum is not long-lived in a dead tree and usually disappears from the above-ground parts of its host within a year of death of the tree. Survival in underground parts, however, may be considerably longer.
In many locations, the most important means of spread of the fungus to new hosts is through the transportation across root grafts formed between diseased and healthy trees. In some areas, all oaks growing in proximity to one another are grafted together and so spread throughout this colony can be very rapid. Above-ground spread is considerably slower and less common.
Vectors
Oak bark beetles, Pseudopityophthorus minutissimus and P. pruinosus (Coleoptera: Scolytidae), are thought to be important factors in the spread of oak wilt in some areas. In the southern parts of the range of oak wilt, these beetles overwinter in all life stages except pupa, whereas in the north, only the larger larvae successfully overwinter. In either case, spores are carried by adult beetles and feeding wounds created in the bark, in twig crotches, leaf axils etc., serve as suitable infection courts for oak wilt. As many as 30% of beetles emerging from some trees have been reported to be carrying the fungus, but the norm is 0.4 - 2.5%.
Sap beetles (Coleoptera: Nitidulidae) are apparently the important group of vectors in the northern reaches of the fungus' range. In Minnesota, Iowa, West Virginia and Wisconsin, these beetles transmit the fungus when they fly from the sporulating mats produced by B. fagacearum to fresh wounds on healthy trees (Juzwik et al.,1985). The fungal mats produce a sweet fruity odour which attracts the insects and, as the beetles crawl across these mats, the sticky fungal spores adhere to them (French & Stienstra, 1980; Davies, 1992). The combination of sporulating fungus, abundance of active insect vectors and wounded trees due to pruning in May and June make this the time of year when most local spread occurs in Minnesota (Juzwik et al., 1985).
Detection & Identification
Symptoms
In the red oak group, symptoms are characterized by a wilting and bronzing of the foliage, starting at the tree top and tips of branches, and spreading rapidly throughout the entire crown. Symptoms develop beginning in May and continue throughout the growing season. Individual leaves turn bronze progressively, from the tip to the base, sometimes leaving a small area of green tissue at the base around the midrib (French & Stienstra, 1980) (figure 3). Leaves in all stages of discolouration, including green leaves, are shed more or less continuously as the disease progresses. Some diffuse staining may be observed in the outermost sapwood, where the fungus has induced the tree to produce gums and tyloses which discolour the wood (French & Stienstra, 1980).
The fungus produces endoconidia in gray or tan mats beneath the bark of infected, dying or dead trees (figure 2). In some trees, more commonly red oaks, mycelial growths within these mats enlarge to form cushion-shaped structures called pressure pads which can exert sufficient pressure to raise and split the bark. A fruity odour which attracts insects is produced by the mats. Trees of all ages may be affected and usually die the same season that the symptoms appear.
Members of the white oak group are more resistant to oak wilt and may live several years before they die. Some may even recover from infection. Some white oaks may carry the fungus with no foliar symptoms of disease. Generally, however, bronzing is less pronounced and defoliation is reduced in the white oaks, frequently occurring only on a few branches of affected trees. Leaves turn brown from the leaf tip toward the leaf base and discolouration patterns resemble those due to normal fall colour changes (French & Stienstra, 1980). Distinct staining or streaking is often observed in the xylem of affected branches of white oaks (figure 4), less frequently in red oaks.
Bur oak susceptibility is intermediate between that of red oaks and that of white oaks; some bur oaks are severely affected and die rapidly, while others die as slowly as white oaks (French & Stienstra, 1980).

Morphology
Wood samples containing xylem may be cultured on malt agar to produce greyish fungal colonies that smell sweet and fruity. Endoconidiophores and endospores are produced in culture. Perithecia appear after 7 - 10 days growth, being flask-shaped and black, measuring 240 - 380 µm in diameter with an erect beak 250 - 450 µm long. Ascospores are hyaline, one-celled, elliptical, 2-3 × 5-10 µm, and exuded in a sticky creamy-white mass.
Detection and inspection methods
Symptoms are variable and not always evident, so laboratory analysis is necessary to confirm infection. Wood samples should be collected from branches (>2 cm diameter) or main stems while the inner bark is still fresh and green (EPPO, 2001). Several small chips of wood, each containing the two or three outer annual rings, should be placed into 2% malt agar, after sterilization in alcohol, and incubated in light or dark for 8 - 10 days at 20 - 25°C prior to examination.
In culture, mycelium appears around the chips after 3 - 5 days. The conidiophores occur in loose clusters, often irregularly distributed on the mycelium. The culture is greyish and a sweet, fruity smell may be detected. A complete morphological description is provided in the EPPO diagnostic protocol (EPPO, 2001). Endoconidiophores and endospores are produced, and if the two mating types are present, perithecia exuding ascospores in a creamy-white mass will also be produced.
Means of Movement and Dispersal
Local spread occurs naturally be means of root grafting between closely spaced trees or through feeding and tunnelling activities of various insects.
Long distance spread could be anticipated to occur as a result of movement in trade of wood products, particularly those with bark attached which could harbour oak bark beetles, or infected nursery stock used for planting. Sporulating fungal mats form on as many as 50% of infected trees in the red oak group, but are extremely rare in white oaks, so the former is considered to be the most likely pathway for human-assisted movement of oak wilt (Liese & Ruetze, 1987). Transportation of firewood from diseased red oaks may similarly result in further distribution of the fungus since fungal mats in the bark may attract beetles which could subsequently carry fungal spores to new locations (Davies, 1992). Diseased red oak firewood is believed to have been the source of the oak wilt infection in three west Texas counties, removed from the main infected area in Texas by 150 or more miles (Davies, 1992). Bretziella fagacearum can be isolated from sawn lumber up to 24 weeks after sawing (Gibbs & French, 1980).
There is no evidence that seeds or foliage of infected hosts serve as a means of movement, either locally or over long distances, for Bretziella fagacearum.
Pest Significance
Economic or Environmental Impact:
Quercus species in the red oak group are usually killed within one year of becoming infected, whereas white oaks frequently recover from infection or take many years to die. Disease severity varies greatly and is worst in the northwest part of its range (Gibbs & French, 1980). Although in many parts of the infested areas of the United States, there is minimal impact on the local forest-based economy, in Texas, Minnesota and Wisconsin, rapid death of oak woods, primarily Quercus fusiformis in the south and Q. ellipsoidalis further north, is reported.
In urban areas where susceptible oaks are abundant, the impact on property or other social values has been significant. In central Texas, for instance, oak wilt has caused considerable decline in urban and rural property values through landscape degradation, shade privation and resulting decline in property values. The Texas Forest Service estimated in 1990 that over 10,000 trees worth millions of dollars were lost in the City of Austin alone (Davies, 1992).
Control:
There is currently no available cure for oak wilt-infected trees so the best approach is to avoid or reduce infection in areas where the disease occurs and prevent its inadvertent introduction to areas that are presently disease-free (French & Stienstra, 1980).
Measures to reduce the number of new infections in an already infested area include:
- surveillance and removal of diseased trees, removal of potential inoculum sources;
- preventing the formation of, or severing existing root grafts between diseased and healthy trees;
- minimizing wounds, including pruning wounds, on healthy trees, especially during the flight period of potential insect vectors.
As all species of oak appear to be most susceptible to infection by insect-disseminated spores in May and early June, it is important to remove any sources of inoculum that are likely to be producing spores during this time (French & Stienstra, 1980). Diseased red oaks are of greater concern as sources of future infections, because B. fagacearum sporulates most abundantly on trees in this group. Red oaks that wilt in July or August may be expected to be producing substantial quantities of spores in the spring of the following year (French & Stienstra, 1980).
Spread through root grafts can be prevented by mechanically trenching around infected trees or stands of trees using specialized equipment, or chemically through the application of Vapam (sodium methyldithiocarbamate) or a similar herbicide which kills trees roots it contacts (French & Stienstra, 1980).
Since healthy trees are most susceptible to infection in spring, pruning should be avoided during this time. Wounds due to storm breakage or other causes should be covered with a wound dressing as soon as possible (French & Stienstra, 1980).
Phytosanitary Measures
Steaming or kiln-drying wood or lumber eliminates Bretziella fagacearum from infected products (Gibbs & French, 1980). The following treatments successfully eliminated B. fagacearum from infected red oak (Quercus rubra, Q. velutina and Q. coccinea) logs:
- compete immersion of the log(s) in hot water at 49°C or higher for at least 12 hours (Jones, 1973);
- compete immersion of the log(s) in hot water at 43°C or higher for at least 48 hours (Jones, 1973);
- compete immersion of the log(s) in hot air at 54°C or higher for at least 24 hours (Jones, 1973);
- compete immersion of the log(s) in hot air at 43°C or higher for at least 48 hours (Jones, 1973).
Methyl bromide fumigation of logs or lumber is also an effective means of eliminating the oak wilt fungus; the following rates have been successfully evaluated in laboratory and field fumigation trials:
- Logs: 240 g/m3 methyl bromide for 3 days (72 hours) at temperatures at or above 5°C (Schmidt et al., 1982)
- Logs: 240 g/m3 methyl bromide for 2 - 3 days (48 - 72 hours) at temperatures of 2 - 5°C or higher (MacDonald et al., 1985)
- Lumber: 240 g/m3 methyl bromide for 2 days (48 hours) at temperatures at or above 5°C (Schmidt, 1982).
Fumigation with methyl bromide at temperatures below 0°C were ineffective even when fumigant concentration and duration of treatment were increased (Schmidt, 1983). The USDA Treatment Manual Schedule T312 - PDF (5000 kb) describes approved rates for treatment of oak logs and lumber for the control of oak wilt disease.
Sulfuryl fluoride is also an effective fumigant for eliminating B. fagacearum in oak logs, and is a suitable alternative to methyl bromide when applied at treatment levels equal to or greater than 27,400 g h/m3 or 280 g/m3 for 72 hours (Woodward & Schmidt, 1995; Schmidt et al., 1997).
Red oak logs are considered a much more likely pathway for entry of oak wilt than are white oak logs (Miller et al., 1985). Spraying a 10% solution of sodium nitrite on the heartwood of oak logs is a simple colour test that can be used to distinguish red from white oaks for the purposes of enforcing different treatment regimes. When a 10% solution of sodium nitrite is sprayed on the heartwood of white oak, a distinct colour change is immediately evident. The sprayed area immediately turns yellowish or brown with an orange tint, and within five minutes is dark greenish or purplish brown to black, fading again over the next several hours (Miller et al. 1985). On red oaks, the same treat results in a similar immediate reaction, a yellowish or brown colouration, but no dramatic darkening follows.
Bibliography
Bretz, T.W. and W.G. Long, 1950. Oak wilt fungus isolated from Chinese chestnuts. Plant Disease Reporter 34: 291.
Davies, C.S., 1992. Environmental Management of Oak Wilt Disease in Central Texas. Environmental Management 16: 323 - 333.
EPPO, 2001. Diagnostic protocols for regulated pests. Ceratocystis fagacearum. OEPP/EPPO Bulletin 31: 41 - 44.
Farr, D.F., G.F. Bills, G.P. Chamuris and A.Y. Rossman, 1989. Fungi on plants and plant products in the United States. APS Press, St. Paul, Minnesota, USA. 1252 pp.
French, D.W. and W.C. Stienstra, 1980. Oak Wilt. Extension Folder 310 - Revised 1980. Agricultural Extension Service, University of Minnesota. 6 pp.
Gibbs, J.N. and D.W. French, 1980. The transmission of oak wilt. USDA Forest Service Research Paper NC-185. 17 pp.
Jones, T.W., 1973. Killing the oak wilt fungus in logs. Forest Products Journal 23: 52 - 54.
Juzwik, J., D.W. French and J. Jeresek, 1985. Overland spread of the oak wilt fungus in Minnesota. J. Arboriculture 11: 323 - 327.
Liese, W. and M. Ruetze, 1987. On the risk of introducing oak wilt on white oak logs from North America. Arboriculture Journal 11: 237 - 244.
MacDonald, W.L., E.L. Schmidt and E.J. Harner, 1985. Methyl bromide eradication of the oak wilt fungus from red and white oak logs. Forest Products Journal 35: 11 - 16.
Miller, R.B., J.T. Quirk and D.J. Christensen, 1985. Identifying white oak logs with sodium nitrite. Forest Products Journal 35: 33 - 38.
Rexrode, C.O. and D. Brown, 1983. Oak Wilt. Forest Insect and Disease Leaflet 29. Northcentral Forest Experiment Station, Minnesota, USA. 5 pp.
Schmidt, E.L., 1982. Methyl bromide eradication of the oak wilt fungus from lumber. International Journal of Wood Preservation 2: 123 - 126.
Schmidt, E.L., 1983. Minimum temperatures for methyl bromide eradication of Ceratocystis fagacearum in red oak log pieces. Plant Disease 67: 1338 - 1339.
Schmidt, E.L., M.M. Ruetze and D.W. French, 1982. Methyl bromide treatment of oak wilt infected logs: laboratory and preliminary field fumigations. Forest Products Journal 32: 46 - 49.
Schmidt, E., J. Juzwik and B. Schneider, 1997. Sulfuryl fluoride fumigation of red oak logs eradicates the oak wilt fungus. Holz als Roh und Werkstoff 55: 315 - 318.
Sinclair, W.A., H.H. Lyon and W. T. Johnson, 1987. Diseases of trees and shrubs. Cornell University Press, Ithaca and London. 575 pp.
Wilson, A.D., 2001. Oak Wilt. A potential threat to southern and western oak forests. J. of Forestry 99: 4 - 11.
Text: Plant Health Risk Assessment Unit.
- Date modified:

